What product(s) are formed when a Grignard reagent prepared from each of the following alkyl halides is
Question:
What product(s) are formed when a Grignard reagent prepared from each of the following alkyl halides is treated with D2O?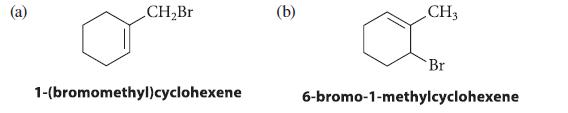
Transcribed Image Text:
CH₂Br 1-(bromomethyl)cyclohexene (b) CH3 Br 6-bromo-1-methylcyclohexene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
b The initially formed Grignard reagent undergoes a rapid al...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What product(s) are formed when a Grignard reagent prepared from the following alkyl halides is treated with D20? CH3 6-bromo-1-methylcyclohexene
-
Predict the major product that is obtained when each of the following alkyl halides is treated with potassium tert- butoxide. Explain your reasoning. (a) -Br (b) OCH3 T CH-CHI
-
What substitution and elimination products (if any)might be obtained when each of the following alkyl halides is treated with sodium methoxide in methanol? (a) methyl iodide (b)...
-
What favors the formation of continuous (dense) cleavage?
-
Working backward from changes in the Buildings and Equipment account The comparative balance sheets of American Airlines show a balance in the Buildings and Equipment account at cost year-end of...
-
Gulf Coast Corporation is a successful oil and gas exploration business in the southwestern United States. At the beginning of 2011, the company made investments in three companies that perform...
-
What statement can be made about the masses of atoms in the above reaction? A. \(m\left({ }_{92}^{235} \mathrm{U} ight)>m\left({ }_{56}^{141} \mathrm{Ba} ight)+m\left({ }_{36}^{92} \mathrm{Kr}...
-
Madero Glass Company manufactures three types of safety plate glass: large, medium, and small. All three products have high demand. Thus, Madero Glass is able to sell all the safety glass that it can...
-
In the world of correctional administration, maintaining discipline and order is of paramount importance. One of the key tools at our disposal is the concept of progressive discipline for employee...
-
In each case, give the structure of a starting material that would give the product shown by MnO 2 oxidation @ CH=O (b) O (c) HOCH-
-
What product(s) are expected when each of the following compounds reacts with one equivalent of NBS in CCl 4 in the presence of light and peroxides? Explain your answers. (a) cyclohexene (b)...
-
If you average 30 mi/h on a 150-mi trip and then return over the same 150 mi at the rate of 50 mi/h, what is your average speed for the trip? Give reasons for your answer.
-
In 2019, Mary Kelly drove her personal car 2,800 miles for business purposes. She also incurred $182 in parking fees and $191 in turnpike tolls connected with her business trips. Compute Marys...
-
Explain the tax treatment oft he following business casualty losses: complete destruction of the property and partial destruction of the property.
-
Identify the six tests concerning rental properties that help to establish a nonrental activity.
-
What relief from passive loss restrictions is provided to help taxpayers deduct losses from rental real estate activities? What are the qualifications?
-
Norm and Pat, a married couple filing a joint return, have the following sources of income: Both Norm and Pat are over 65 years of age. Determine their taxable income. Wages Interest income Dividend...
-
Some New York Stock Exchange analysts believe that 24-hour trading on the stock exchange is the wave of the future. As an initial test of this idea, the New York Stock Exchange opened two after-hour...
-
A statistical study shows that the fraction of television sets of a certain brand that are still in service after x years is given by f (x) = e-0.15x. (a) What fraction of the sets are still in...
-
How would the NMR spectrum of ethyl fluoride differ from that of ethyl chloride?
-
Describe in detail what changes you would expect to see in the NMR resonance of the methyl group as 1-chloro-l-methylcyclohexane is cooled from room temperature to very low temperature.
-
The proton-decoupled 13C NMR spectra of 3-heptanol (A) and 4-heptanol (B) are given in Fig. 13.22 on page 626. Indicate which compound goes with each spectrum, and explain your reasoning. Fig. 13.22...
-
describe the social contract implied or guaranteed to citizens by the establishment of immigration laws of discrimianition, saftey and education ?
-
Question No 2: Among a large group of patients recovering from shoulder injuries, it is found that 25% visit both a physical therapist and a chiropractor, and 85% visit at least one of these. The...
-
You wish to accumulate $ 1 4 1 9 7 in 9 years. Payments are made at the end of every six - month period into an account earning 7 . 4 % compounded semi - annually. Find the required payment amount to...

Study smarter with the SolutionInn App


