Why is base (NaOH or KOH) added as a second step in each of the reactions shown
Question:
Why is base (NaOH or KOH) added as a second step in each of the reactions shown in Eqs. 23.23 and 23.24?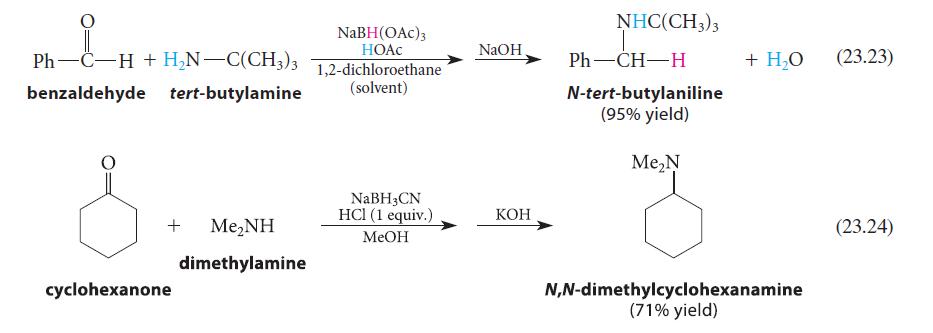
Transcribed Image Text:
Ph—C−H+H_N—C(CH3)3 benzaldehyde tert-butylamine + Me,NH dimethylamine cyclohexanone NaBH(OAC) 3 HOAC 1,2-dichloroethane (solvent) NaBH 3 CN HCl (1 equiv.) MeOH NaOH KOH NHC(CH3)3 Ph—CH—H N-tert-butylaniline (95% yield) Me₂N + H₂O N,N-dimethylcyclohexanamine (71% yield) (23.23) (23.24)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Under the acidic conditions shown in Eqs 2323 acetic acid and 2324 HCI the initial product w...View the full answer

Answered By

Dorcas Juliet
I am a proficient tutor and writer with over 4 years experience, I can deliver A+ works in all fields related to business and economics subject. Kindly hire me for excellent papers
4.70+
10+ Reviews
51+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In April 1992, EuroDisney SCA opened its doors to European visitors. Located by the river Marne some 20 miles east of Paris, it was designed to be the biggest and most lavish theme park that Walt...
-
Each of the reactions shown involves nucleophilic substitution. The product of reaction (a) is an isomer of the product of reaction (b). What kind of isomer? By what mechanism does nucleophilic...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Sparkle is one firm of many in the market for toothpaste, which is in long-run equilibrium. a. Draw a diagram showing Sparkles demand curve, marginal-revenue curve, average total- cost curve, and...
-
What are generally accepted accounting principles (GAAP)?
-
You are beginning your first audit of Suncoast Network Systems, Inc., a company that develops network security software. During your investigation of internal controls you learn that the companys...
-
Reconsider Problem 56. Determine which oven should be purchased based on an incremental annual worth analysis. Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The...
-
Nonmonetary Exchanges Holyfield Corporation wishes to exchange a machine used in its operations. Holyfield has received the following offers from other companies in the industry. 1. Dorsett Company...
-
(d) What is the value of "j" after the execution of the partial code in Figure 4.2? Explain your answer. int j = 10; for (int i = 0; 0; i
-
Quaternization (Sec. 23.6) does not occur in the reactions shown in Eqs. 23.26 and 23.27. Explain. NH + HC=0 NaBH3CN HOAC CH3CN/HO EtNHCH,Ph + H,C=O benzylethylamine formaldehyde NaBH3CN CH3CN/HO...
-
Provide a reaction mechanism for step 1, formation of the imine, in Eq. 23.22. EtNH + H3C-C-CH3 acetone -HO [HC- NEt H3C-C-CH3 an imine (not isolated) H, Pt 30 psi EtOH NHET H3C-CH-CH3 (23.22)...
-
Crayola, LLC, is a profitable, wholly owned subsidiary of Hallmark Cards of Kansas City, Missouri. The companys world headquarters are located in Easton, Pennsylvania; departments there include house...
-
Glass is a hard, transparent material which is used to make windows, bottles, and other objects. It is made by cooling molten ingredients such as silica sand with sufficient rapidity to prevent the...
-
1 kg of aluminum requires 887 J (Joule) to raise its temperature by 1 K (Kelvin). The specific heat of aluminum is expressed as (J/(kg*K)). For a 100 g aluminum bar, how much energy is needed to...
-
Refer to the policies and procedures Referring to the Education and Care Services National Regulations A) Identify the Regulation that is relevant for the medical condition asthma? b)Provide one (1)...
-
3. Consider non-lifting flow over circular cylinder with radius = 0.5 m and freestream velocity = 3 m/s. Determine the velocity and the pressure coefficient at the following locations on the surface...
-
ABC Mining has undertaken a Pre-Feasibility Study (PFS) for its ABC Project and is preparing a Public Report to the ASX reporting the resulting Probable Ore Reserves for the first time. ABC and the...
-
Describe the principal steps in the design phase. What are the major deliverables?
-
In Problems 1522, find the principal needed now to get each amount; that is, find the present value. To get $750 after 2 years at 2.5% compounded quarterly.
-
Acid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.
-
Acid-catalyzed dehydration of either 2-methyl-1-butanol or 3-methyl-1-butanol gives 2-methyl-2-butene as the major product. Write plausible mechanisms that explain these results.
-
When the compound called isoborneol is heated with 9 M sulfuric acid, the product of the reaction is the compound called camphene and not bornylene, as one might expect. Using models to assist you,...
-
Pension data for Sterling Properties include the following: ($ in thousands) $ 127 Service cost, 2024 Projected benefit obligation, January 1, 2024 Plan assets (fair value), January 1, 2024 Prior...
-
On January 1, 2024, Alamar Corporation acquired a 35 percent interest in Burks, Incorporated, for $235,000. On that date, Burks's balance sheet disclosed net assets with both a fair and book value of...
-
PA12-4 Preparing and Interpreting a Statement of Cash Flows (Indirect Method) Heads Up Company was started several years ago by two hockey instructors. The company comparative balance sheets and...

Study smarter with the SolutionInn App


