Suggest a structure for a constitutional isomer of the following compound that should have greater water solubility,
Question:
Suggest a structure for a constitutional isomer of the following compound that should have greater water solubility, and explain your reasoning. The structure should not be an enol, because enols are not stable. 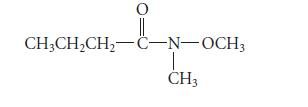
Transcribed Image Text:
O || CH3CH₂CH₂-C-N-OCH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
The compound shown in the image is Nmethylpropanamide A constitutional isomer of this compound ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The UV spectrum of 1-phenylprop-2-en-1-ol shows an intense absorption at 220 nm and a weaker absorption at 258 nm. When this compound is treated with dilute sulfuric acid, it rearranges to an isomer...
-
our class. The word count of the text that includes your "own words" should be at least 500 words (about 700 max.). The text should be typed double-spaced using a 12-point font size in Times New...
-
(a) Two amides are constitutional isomers and have the formula C 4 H 9 NO, and each contains an isopropyl group as part of its structure. Give structures for these two isomeric amides. (b) Draw the...
-
Cost-Volume-Profit (CVP) analysis can be used to determine the effect of changes in costs and volumes on a company's net profits. This project assignment assumes that you are newly hired as an...
-
Identify the questions you should ask to anticipate your audiences reaction.
-
Tab Corporation decided to establish Collon Company as a wholly owned subsidiary by transferring some of its existing assets and liabilities to the new entity. In exchange, Collon issued Tab 30,000...
-
The summary statistics for a certain set of points are: n = 20, se = 4.65, (x x)2 = 118.26, and b1 = 1.62. Assume the conditions of the linear model hold. A 99% confidence interval for 1 will be...
-
Mountain View Manufacturing Co. maintains the following accounts in the general ledger: Materials, Work in Process, Factory Overhead, and Accounts Payable. On July 1, the materials account had a...
-
Question 2 (11 marks) Maria Makeba, CFA wants to construct a portfolio which includes government securities and equity exposure. She intends to use Treasury bills and an index fund tracking the JSE...
-
Vitamins can be classified as fat-soluble or watersoluble. Fat-soluble vitamins can be stored in fatty tissues, whereas water-soluble vitamins can be excreted in the urine. (a) The structures of some...
-
When salad oil is mixed with water and shaken, two layers quickly separate, the oily layer on top and the water layer on the bottom. When an egg yolk (which is rich in lecithin, a phospholipid) is...
-
Compute the Z score for Giant Motors Corporation (GMC) given the following information for year-end 2012: At what share price would GMC have a Z score equal to 3.00? How does the future look for GMC?...
-
General Energy Storage Systems (GESS) was founded in 2012 by Ian Redoks, a Ph.D. candidate in physics who was interested in "outside-the-box" solutions to the problem of storing electrical energy....
-
General Energy Storage Systems (GESS) was founded in 2012 by Ian Redoks, a Ph.D. candidate in physics who was interested in "outside-the-box" solutions to the problem of storing electrical energy....
-
INNOVATIVE PRINT SYSTEMS: PRODUCTION PLANNING AND SHIPPING Innovative Print Systems is a technology company that produces 3D printers. Four different types of printers (differing mainly by using...
-
As Marketing Manager think of a print ad you saw recently. Briefly describe the ad? Why is the following question the most important when you are evaluating your print ad? Is the brand or...
-
A retail store you own requires the tenant to pay percentage rent of 5% over their break point of $1 million per year. Their sales have been $1.35 million per year and you have been investing the...
-
A minibar in a hotel room generally provides an impression that the hotel is more upscale. PKF Hospitality research reported that minibars provide a mean annual revenue of $368 per hotel room (USA...
-
Periwinkle Company is a multinational organization. Its Parts Division is located in Lavender Land, while its Assembly Division is located in North Orchid. During the current year Periwinkle Companys...
-
Provide names for thesecompounds: a) CH;CH,CH,CH,CH,CH b) CH f) CH,CCH,CCH, d) CI g) h)
-
Draw the structures for these compounds: (a) (Z)-Oct-3-en-2-one (b) 3-Ethylheptanal (c) 2, 4-Pentadienal (d) 3, 4-Dimethylbenzaldehyde (e) 1-Phenyl-1-propanone (f) 2, 2, 6, 6-Ttramythleyelohexanone
-
Explain which the most acidic hydrogen's in these compoundsare c) CH,CCH,CH, b) PHCH CCH3
-
The following financial statements and additional information are reported. IKIBAN INCORPORATED Comparative Balance Sheets At June 30 Assets Cash Accounts receivable, net Prepaid expenses Inventory...
-
Liquidation Basis of Accounting: Working Backwards Guadeloupe Company is in the process of liquidating, and is using the liquidation basis of accounting. Its beginning and ending statements of net...
-
No indirect materials were issued from inventory during the period. The controller had just allocated the underapplied overhead to Cost of Goods Sold, Finished Goods Inventory, and Work-in-Process...

Study smarter with the SolutionInn App


