Use the known stereochemistry of the starting alkene (from this section) to assign the stereochemical configuration of
Question:
Use the known stereochemistry of the starting alkene (from this section) to assign the stereochemical configuration of the product, which was found by experiment to be levorotatory.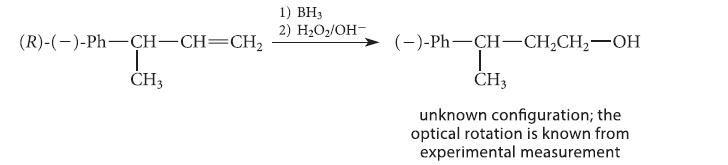
Transcribed Image Text:
(R)-(-)-Ph-CH-CH=CH₂ | CH3 1) BH3 2) H₂O₂/OH- (-)-Ph-CH-CH₂CH₂-OH I CH3 unknown configuration; the optical rotation is known from experimental measurement
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Begin with the perspective structure of the reactant in ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain how you would use the alkene starting material in Problem 6.13 to determine the absolute configuration of the dextrorotatory enantiomer of the following hydrocarbon: Indicate what reaction...
-
From time to time courts are called on to determine the enforceability of an arbitration clause contained in a contract. One such case was DiMercurio v.Sphere Drake Insurance, 202 F. 3d 71 (1st Cir....
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Accurate Job Costing must be done on three levels. Which of the following is not one of these levels? Tracking and controlling costs during jobs Tracking gross profit each month Filing records on...
-
Assuming the guards did a bad job assisting the passenger, was the railroad liable for the injuries to Ms. Palsgraff?
-
Porter Company purchased 60 percent ownership of Temple Corporation on January 1, 20X1, at underlying book value. At that date, the fair value of the noncontrolling interest was equal to 40 percent...
-
Anthonys Consulting, Inc., started business in 2010 with total assets of \($32,000\) and total liabilities of \($14,000\). At the end of 2010, Anthonys total assets were \($42,000\), and total...
-
1. As a digital retailer, how does Alibaba provide value to Chinese consumers? What sets of values are unique to the Chinese market? 2. Given that Alibaba does not own or distribute any of the...
-
Differential equations In Exercises 27-42, find the solution y = y(x) to the given initial-value problem. On what interval is the solution valid? (Note that exercises involving differential equations...
-
Explain why the following compound has two meso stereoisomers. | | | H3C-CH-CH-CH-CH3
-
A chemist has developed a new synthesis of ibuprofen and has reported that she has prepared the (S)-(1)-enantiomer in 90% EE, and that this material has a measured specific rotation of 151.7 degrees...
-
Find the limit. 1 - tan x lim xT/4 sin x cos x
-
A London based high-tech firm produces component parts that are sold to cellular phone manufacturers as well as a diverse set of customers across several different industry sectors. The firm uses a...
-
In which phase of risk management does the valuation of scenarios occur. Explain in details and provide example.
-
what extent do organizational structures and systems, including hierarchical arrangements, matrix structures, and agile methodologies, impact employee motivation, job satisfaction, and overall...
-
Rooney Manufacturing Company was started on January 1, year 1, when it acquired $83,000 cash by issuing common stock. Rooney immediately purchased office furniture and manufacturing equipment costing...
-
Bruce is considering the purchase of a restaurant named Hard Rock Hollywood. With the help of his accountant, Bruce projects the net cash flows (cash inflows less cash outflows) from the restaurant...
-
Suppose that we have a sample space with five equally likely experimental outcomes: E1, E2, E3, E4, E5. Let A = {E1, E2} B = {E3, E4} C = {E2, E3, E5} a. Find P(A), P(B), and P(C). b. Find P(A ( B)....
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
Rank the following halides in order of their reactivity in the Williamson synthesis: (a) Bromo ethane, 2-bromopropane, bromo benzene (b) Chloro ethane, bromo ethane, 1-iodopropene
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Write the mechanism of the acid-catalyzed cleavage of tert-butyl cyclohexyl ether to yield Cyclohexanol and 2-methylpropene.
-
For E>0, there is a corresponding N such that value of L will be 2 Sn L N. The 4"+3
-
Use the appropriate test, choose from using Geometric Series Test or Test for Divergence. Determine whether the series is convergent or divergent. If it is convergent, find its sum. M 2 +4 en
-
For each of the following, find x in terms of y: 5 4x + 3y = 2x + 21y

Study smarter with the SolutionInn App


