What is the configuration of the following stereoisomer of 3-methyl-2-pentene? (The numbers are for reference in the
Question:
What is the configuration of the following stereoisomer of 3-methyl-2-pentene? (The numbers are for reference in the solution.)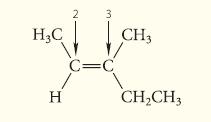
Transcribed Image Text:
H3C Н 2 3 c=c CH3 CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Assign the relative priorities of the two groups attached to each carbon ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The stereoisomer of 1-iodo-2-methylbutane with the S configuration rotates the plane of polarized light counterclockwise. The following reaction results in an alcohol that rotates the plane of...
-
For many centuries, the Chinese have used extracts from a group of herbs known as ephedra to treat asthma. Chemists have been able to isolate a compound from these herbs, which they named ephedrine,...
-
What is the configuration (E or Z) of the following alkene? (The numbers and letters are for reference in the solution.) H 2 HC C=C 02 CHCHCBrCH3 61 62 CHCHCH3 T CH3 6,6-dibromo-3-isobutyl-2-heptene
-
Unlike other sciences, the nature and value of philosophy is derived from reflection versus testing, measuring, or examining. O True O False 4
-
If you are persuaded by the Collegiate Times position, please answer the following questions:
-
Ricks is a popular restaurant for fine dining. The owner and chef, Rick Goetz, is pleased with his success and is now considering expanding his existing restaurant or perhaps opening a second...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
(Entries for Bond TransactionsStraight-Line) Foreman Company issued $800,000 of 10%, 20-year bonds on January 1, 2011, at 102. Interest is payable semiannually on July 1 and January 1. Foreman...
-
To mix plaster for a dental model, 45 milliliters (mL) of water are used for 100 grams (g) of plaster. How many mL of water should be used for 200 g of plaster? . A cardiopulmonary resuscitation...
-
Give the structure of: (a) (E)-4-allyl-1,5-octadiene (b) (2E,7Z)-5-[(E)-1-propenyl]-2,7-nonadiene Be sure to read Study Guide Link 4.2 if you have difficulty with this problem.
-
Name the following compounds. Ignore double-bond stereochemistry. (a) Me Me (b) CH3CHCH=CHCHCHCH3
-
Everett has just received $500 as a birthday gift and has decided to spend it on DVDs and video games. He has determined that the utility (satisfaction) derived from the purchase of x DVDs and y...
-
Imagine that you are the marketing director of an advertising agency and you must present a proposal on the services provided by the agency. The client owns a sports equipment store and wants to...
-
Woody Company has the following balances for the current month: Indirect materials used $1,799 Direct labor 6,673 Sales salaries 11,504 Indirect labor 1,871 Production manager's salary 11,080...
-
Your CFO has supplied you with the following information. Current product standard costs are as follows: Selling price per unit: $5,000 $1,400/unit direct material $400/unit direct labor $200/unit...
-
how to calculate valuation of a company based off statement of cash flow, balance sheet, income statement, and stock history ?Explain
-
North Division has the following information: Sales $1190000 Variable expenses 649000 Fixed expenses 620000 If this division is eliminated, the fixed expenses can be allocated to the company's other...
-
How will each of the following changes in demand and/or supply affect equilibrium price and equilibrium quantity in a competitive market; that is, do price and quantity rise, fall, or remain...
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
Identify each of the colored positions?red, blue, and green?as axial or equatorial. Then carry out a ring-flip, and show the new positions occupied by each color, Ring-flip
-
What is the energy difference between the axial and equatorial conformations of cyclohexanol (hydroxycyclohexane)?
-
Why do you suppose an axial cyano (CN) substituent causes practically no 1, 3-diaxial steric strain (0.4 kJ/mol)? Use molecular models to help with your answer.
-
Encik Jeff, a Canadian citizen, left Malaysia permanently on 3 1 October 2 0 2 2 . The record of Encik Jeff s stays in Malaysia have been as follows: Year 2 0 1 6 , He stay in Malaysia 2 0 December...
-
In Canada, the federal tax rate you have to pay depends on your income bracket. - 1 5 % on the first $ 5 3 , 3 5 8 of taxable income. - 2 0 . 5 % on taxable income over $ 5 3 , 3 5 9 up to $ 1 0 6 ,...
-
Exercise 4. Company XYZ is evaluating the feasibility of manufacturing a new product. The plant it owns has the capacity to produce 1,000,000 units of the product. Fixed costs for this initiative...

Study smarter with the SolutionInn App


