When 1,3,5-cyclooctatriene, A, is heated to 80100C, it comes to equilibrium with an isomeric compound B. Treatment
Question:
When 1,3,5-cyclooctatriene, A, is heated to 80–100°C, it comes to equilibrium with an isomeric compound B. Treatment of the mixture of A and B with CH3O2C—C≡C—CO2CH3 gives a compound C, which, when heated to 200°C for 20 minutes, gives dimethyl phthalate and cyclobutene. Identify compounds B and C, and explain what reactions have occurred.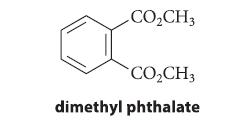
Transcribed Image Text:
CO₂CH3 CO₂CH3 dimethyl phthalate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Take a cue from the solution to Problem 2831a on p 1031XR o...View the full answer

Answered By

Loise Ndungu
I have five years of experience as a writer. As I embark on writing your papers from the prologue to the epilogue, my enthusiasm is driven by the importance of producing a quality product. I put premium product delivery as my top priority, as this is what my clients are seeking and what makes me different from other writers. My goal is to craft a masterpiece each time I embark on a freelance work task! I'm a freelance writer who provides his customers with outstanding and remarkable custom writings on various subjects. Let's work together for perfect grades.
4.90+
78+ Reviews
224+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound A (C 11 H 14 O 3 ) is insoluble in base and gives an isomeric compound B when heated strongly. Compound B gives a sodium salt when treated with NaOH. Treatment of the sodium salt of B with...
-
When compound A is irradiated with ultraviolet light for 115 hours in pentane, an isomeric compound B is obtained that decolorizes bromine in CH2C12 and reacts with ozone to give, after the usual...
-
If isomer A is heated to about 100 °C, a mixture of isomers A and B is formed. Explain why there is no trace of isomer C or D. CDa CHs CD2 C6H5 CD3 Hs CD3 CH; 100 C CH3 CH CH C%H5 CHs
-
Phoenix Corp. faltered in the recent recession but is recovering. Free cash flow has grown rapidly. Forecasts made in 2016 are as follows. Phoenix's recovery will be complete by 2021, and there will...
-
Trotman Company had three intangible assets at the end of 2012 (end of the accounting year): (a) Computer software and Web development technology purchased on January 1, 2011, for $70,000. The...
-
For each of these three cases, determine the missing amounts indicated by question marks. Case 2 Case 1 $7 19,480 Case 3 $7 34,000 18,000 86,000 38,000 Sales Finished goods inventory, beginning...
-
On April 23, 2014, Calvin Loyer admitted his wife, Edeltrud Loyer, to a nursing home administered by Signature Healthcare. During the admissions process, Calvin signed an arbitration agreement...
-
Brockman Guitar Company is in the business of manufacturing top-quality, steel-string folk guitars. In recent years the company has experienced working capital problems resulting from the procurement...
-
On a private flight with capacity for 150 people, $800,000 per person is charged, plus $25,000 for each unsold seat on the plane. How many people must travel on the plane and what will be the price...
-
The reaction in Fig. P28.41 occurs as a sequence of two pericyclic reactions. Identify the intermediate A, and describe the two reactions. CH3 +BO_i_c_c_i_OF EtO-C- a-phellandrene Figure P28.41 OEt...
-
Classify the following sigmatropic reaction, give the curved- arrow notation, and show that the stereochemistry is that expected for a thermal concerted reaction. (This reaction, discovered by Prof....
-
Consider a competitive market served by many domestic and foreign firms. The domestic demand for these firms product is Qd 600 2P. The supply function of the domestic firms is QSD = 200 P, while that...
-
1. With the advantages and disadvantages of using a systems review committee in mind, when are systems requests better reviewed by a committee, and when are requests better reviewed by an individual....
-
Given that Na2O and SiO2 combine to form the silicate Na2SiO3, use the data below to show that the calcination of NaCO3 is only favourable in the presence of enough silica to form the silicate. All...
-
The Balanced Scorecard offers many advantages as compared to traditional management by objectives. It provides a thorough perspective of an institution's effectiveness to start. However, The Balanced...
-
The uniform 20-kg slender bar AC shown in Figure Q5 rotates in a vertical plane about the pin at B. The ideal spring AD has a spring constant k = 15 N/m and an un-deformed length Lo = 2 m. The bar,...
-
why Rowlandson work is important part of establishing an American literary tradition. You must use specific examples in your response.?
-
A restaurant converts inputs into substantially different outputs, a key characteristic of a manufacturing firm. Yet, most would classify a restaurant as a service firm. How would you classify a...
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Given its mission of providing information to the world, should Google censor searches in China?
-
Estimate each of the bond angles and order the bond lengths (smallest first) for each of the following molecule. (a) a) (b) d) Br:
-
(a) Give the H-C=O bond angle in methyl formate. (b) One dihedral angle in methyl formate relates the plane containing the O=C-O bonds to the plane containing the C-O-C bonds. Sketch two structures...
-
The compound benzenc has only one type of carbon-carbon bond, and this bond has a length intermediate between that of a single bond and a double bond. Draw a resonance structure of benzene that,...
-
Your examination of Procter & Gamble's fiscal 2023 liquidity and current ratio is quite perceptive. It's true that the current ratio being less than 1 indicates P&G's current assets are less than its...
-
The stocks that performed the best this week were Disney (DIS) and Southwest Airlines (LUV). Disney's overall rate of return jumped to 23.57% this week, up significantly from 7.02% two weeks prior....
-
ACC-112 M Question 1 Ch 910 1-4-Com X On January 2, 20x1, Valentina x Romheducation.com/ext/map/index.html? con-con&external browser-08launchUrl=https%253A%252F%252Flms.mheducation.com6252Fm Student...

Study smarter with the SolutionInn App


