Which of the following ions require(s) dihedral angles to specify its structure completely? Explain. + HC-NH3 A
Question:
Which of the following ions require(s) dihedral angles to specify its structure completely? Explain.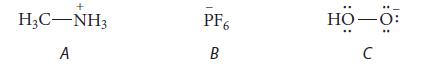
Transcribed Image Text:
+ H₂C-NH3 A PF6 B HO-O: C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
A molecule contains dihedral angles only if it contains at least four atoms connecte...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following ions will always be a spectator ion in a precipitation reaction? (a) CI- (b) NO3- (c) NH4+ (d) S2- (e) SO42- Explain briefly.
-
Which of the following ions possess a noble gas electron configuration? (a) K+ (d) O- (b) He+ (e) F- (c) H- (f) Ca2+
-
Which of the following ions have noble gas electron configurations? a. Fe2+, Fe3+, Sc3+, Co3+ c. Pu4+, Ce4+, Ti4+ b. Tl+, Te2-, Cr3+ d. Ba2+, Pt2+, Mn2+
-
How many moles of carbon dioxide, CO2, are in a 22 gram sample of the compound?
-
Factoring and Outlining a Problem Japan Airlines has asked your company, Connections International, to prepare a proposal for a training school for tour operators. JAL wants to know whether Burbank...
-
Determine the amount of gain or loss realized and the amount of gain or loss to be recognized in each of the following dispositions: a. Jorge owns 800 shares of Archer Company stock. He had purchased...
-
List three ways to get input from the console and convert that input to the desired data type.
-
On January 1, 2012, Moline Company had Accounts Receivable $154,000; Notes Receivable of $12,000; and Allowance for Doubtful Accounts of $13,200. The note receivable is from Hartwig Company. It is a...
-
Regarding 1031 exchanges, which of these statements regarding debt load in an exchanged property is true? Unset starred question The 1031 exchange doesn't address debt load. The debt load for the new...
-
(a) Draw a resonance structure for the allyl anion that shows, along with the following structure, that the two CH 2 carbons are equivalent and indistinguishable. (b) According to the resonance...
-
Use molecular orbital theory to explain why He 2 does not exist. The molecular orbitals of He 2 are formed in the same way as those of H 2 .
-
Consider a parallel-plate capacitor with an area of 1.0 cm 2 , with a plate spacing of 0.20 mm, and filled with mica. At what voltage will this capacitor exhibit dielectric breakdown?
-
Company : Walmart Based on the company please provide the below topics answers in detail. Customer Handling & Store Environment : 1.Advertistemnt 2. Direct selling 3.Sales promotion 4. Publicity...
-
Ethics Committee Baby Charlie and End-Of-Life Decisions Case Study, The Tracks We Leave: Ethics and Management Dilemmas in Health Care, What laws and ethical principles and theories apply, Name,,...
-
On December 7, 2020, Xpeng (XPEV), a Chinese-based electric car maker, filed to raise more money by selling shares to the public. This is called______ and will occur in the _______.
-
1. Drawing upon the Job Analysis of Library Student Assistant, identify and discuss what you deem to be suitable criterion measures. Next, identify and describe potential challenges that may be...
-
What is the tax treatment of interest on U.S. government bonds? How are the life insurance proceeds taxed? What is the tax treatment of debt cancellation?
-
Resource consumption per person in the United States is either flat or falling, depending on the resource. Yet living standards are rising due to improvements in technology that allow more output to...
-
The trade-off theory relies on the threat of financial distress. But why should a public corporation ever have to land in financial distress? According to the theory, the firm should operate at the...
-
Show the products of thesereactions: + HCI b) + HF + HI CH3 + HBr CH3 + HCI CH,CH, (p)
-
Show the products of thesereactions: CH3 HBr a) CH,CHCH-CH2 HBr b) PhC CH 2 HBr
-
Show the products of thesereactions: H,SO,, H,SO4 b) + H,0 + H,0 H,SO. + HO
-
Explain the purpose of statement of changes in equity. Also discuss the presentation format of the same statement.
-
In considering these enhancements to the COSO Framework, what specific challenges do you foresee in implementing ethical risk assessment and fostering a culture of integrity within organizations,...
-
A columnist on barrons.com notes that "all possible knowledge about the 500 biggest stocks is fully discounted in their prices." Therefore, he concludes, "passive ownership of that broad index beats...

Study smarter with the SolutionInn App


