Show the products of thesereactions: + HCI b) + HF + HI CH3 + HBr CH3 +
Question:
Show the products of thesereactions:
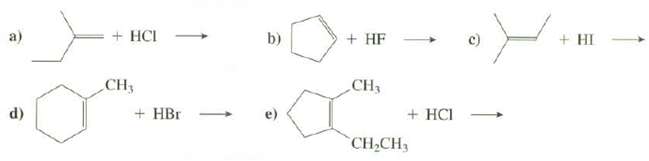
Transcribed Image Text:
+ HCI b) + HF + HI CH3 + HBr CH3 + HCI CH,CH, (p)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
b F d CH3 ...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Answer the Multple Choice Questions and the code for problem 6in the end PROBLEM 1: General UNIX 1. What is UNIX? a) an operating system b) a text editor c) programming language d) software program...
-
Sustainability involves more than just the impact of actions on the environment. The triple bottom line recognizes that a company has to measure its impact on its triple bottom line for its long-term...
-
When is a two-sided message likely to be more effective than a one-sided message?
-
What is the difference between V&V?
-
On January 4, 2010, Dunbar Company purchased, on credit, 2,000 television sets at $500 each. Terms of the purchase were 2/10, n/30. Dunbar paid for 20% of these sets on January 13 and the remaining...
-
Determine the maximum average shear stress developed in each 5/8" diamter bolt: 10a) Is this an example of single or double shear? 10b) If the bolt is an A325 with threads excluded, is it sufficient?...
-
You wish to analyze the pedestrian traffic that passes a given store in a major shopping center. You are interested in determining how many shoppers pass by this store, and you would like to classify...
-
Show the structure of the carbocations that are formed in the reaction of HBr with 2-hexena and explain why two products are formed.
-
Show the products of thesereactions: CH3 HBr a) CH,CHCH-CH2 HBr b) PhC CH 2 HBr
-
For the following exercises, use a graphing utility to graph each pair of polar equations on a domain of [0,4] and then explain the differences shown in the graphs. r = 2sin (/2), r = sin (/2)
-
What European countries originally dominated Canada and the United States during the colonial era, and what are some of the impacts of each of these colonial empires on the political systems and...
-
How are the physical landscapes of the Canadian Shield different from the Appalachian Uplands in Quebecand what geomorphic processes helped shape these two landform regions?
-
How much must a business pay for a fire insurance policy with a face value $750,000, if the annual premium rate is $0.23 per $1,000?
-
A homeowners policy from the RESTEZ Insurance Company costs $3.50 per unit of coverage. Each unit consists of $1,000 for the structure of the house and $200 for contents. Each additional $100 of...
-
How does evidence from color deficiency appear to support the opponent-process channels concept. What are the three basic properties of light?
-
When the bulb of an ideal gas thermometer is immersed in water at the triple point, the height of the mercury column above the reference mark is \(760 \mathrm{~mm}\). The same thermometer is then...
-
Which of the following is FALSE regarding the purchasing power parity (PPP). a. The PPP is a manifestation of the law of one price b. The PPP says that a country with a higher expected inflation can...
-
Order the following solids (ad) from least soluble to most soluble. Ignore any potential reactions of the ions with water. a. AgCl Kp = 1.6 10-10 Ksp = 1.6 10- b. AgS c. CaF d. CuS Ksp = 4.0 10-...
-
How might the following amines be prepared using reductive amination reactions? Show all precursors if more than one ispossible. CH (b) NHCH2CH3 (c) (a) CHCH2NHCHH -NHCH3
-
How could you prepare the following amine using a reductive aminationreaction?
-
How would you prepare the following amines, using both Hofmann and Curtius rearrangements on a carboxylic acidderivative? CH (b) (a) NH2 CH2CH2NH2 CH
-
Convert the following numbers (Show the steps of your work): a. Ox23734 Binary: Decimal: b. Ob01100011 Hex: Decimal: c. 12345 Hex: Binary:
-
Find the total number of compounding periods and the interest rate per period for the investment. Nominal Term of Investment Interest (Annual) Rate (%) Compounded Compounding Periods Rate per Period...
-
A company applies overhead at a rate of 160% of direct labor cost. Actual overhead cost for the current period is $1,006,000, and direct labor cost is $590,000. Determine whether there is over- or...

Study smarter with the SolutionInn App


