Which of the non-hydrogen atoms in each of the following species has a complete octet? What is
Question:
Which of the non-hydrogen atoms in each of the following species has a complete octet? What is the formal charge on each? Assume all unshared valence electrons are shown.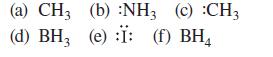
Transcribed Image Text:
(a) CH3 (b) :NH3 (c) :CH3 (d) BH, (e) :Ï: (f) BH4 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The formal charge on all the hydrogens is 0 For the other atoms a Carbon ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the atoms in each of the following species has a complete octet? What is the formal charge on each? Assume all unshared valence electrons are shown. (a) CH (b) :NH3 (c) :CH3 (d) BH3 (e) :T:...
-
Below are the equations for four impulse responses. The plot below shows four step responses. Using the Laplace convolution technique, which impulse response goes with which step response (A, B, C,...
-
Give the formal charge on each atom and the net charge on each species in the following structures. All unshared valence electrons are shown. (a) (b) (c) CH3 trimethylamine oxide methylene :Cl-o:...
-
Sunblessed Juice Company sells bags of oranges and cartons of orange juice. Sunblessed grades oranges on a scale of 1 (poor) to 10 (excellent). At present, Sunblessed has 100,000 pounds of grade 9...
-
Drawing a Line Chart Your Task. Prepare a line chart showing the sales of Sidekick Athletic Shoes, Inc., for these years: 2008, $6.7 million; 2007, $5.4 million; 2006, $3.2 million; 2005, $2.1...
-
In general, taxpayers want to depreciate property as rapidly as possible. Under what circumstances might a taxpayer not want to use accelerated depreciation? How can this be done under MACRS?
-
Use information from Section 6.7 to estimate which form of electromagnetic radiation is the lowest energy ionizing radiation. Data from section 6.7 When we first introduced the concept of the...
-
Leaky Pipe, a local retailer of plumbing supplies, faces demand for one of Its SKUs at a constant rate of 30,000 Units per year. It costs Leaky Pipe $10 to process an order to replenish stock and $1...
-
(a) Consider the recurrence below defined for n 0. 3 T(n): if n = 0 7T (n - 1) +12 if n > 0 Give a closed-form solution to the recurrence. You only have to give the solution. You do not need to show...
-
Draw one Lewis structure for each of the following compounds; show all unshared electron pairs. None of the atoms in the compounds bears a formal charge, and all atoms have octets (hydrogens have...
-
(a) Construct a hybrid orbital picture for the water molecule using oxygen sp 3 hybrid orbitals. (b) Predict any departures from tetrahedral geometry that you might expect from the presence of two...
-
How are persuasive messages different from routine messages?
-
The COVID death toll in the US has reached an incomprehensible number of more then 1 million deaths. With benefit of hindsight assume you decided to form a health care consulting company with the...
-
You found your dream house. It will cost you $250000 and you will put down $40000 as a down payment. If you finance the reminder of the cost with a 30-year 5.0% mortgage, what will your monthly...
-
Define clustering, make sure to provide at least two scholarly and peer-reviewed journal or book references as support. Use APA citation, Do a little digging. Make sure to explain the following...
-
In three hundred words and two references: Respond to each of the following as you think about the changes you have seen in your own community over the past months in response to the COVID 19...
-
Part 1 of Question: What is your professional code? Analyze your code in light of the list of things public administrators "should" do according to Svara: Public administrators should: 1. Be...
-
Aquaculture is the growing of fish, shrimp, and other seafood in enclosed cages or ponds. The cages and ponds not only keep the seafood from swimming away but also provide aquaculturalists with...
-
Refer to the information from Exercise 22-19. Use the information to determine the (1) Weighted average contribution margin , (2) Break-even point in units, and (3) Number of units of each product...
-
Show how these compounds could be synthesized from alkylhalides: a) Heptane b) PHCH,CH,CH,NH, c) PHCH,C=CH d) e) HO, OH f) g) H.C
-
Show how this synthesis might beaccomplished: Br CH3 CH3 HO from "CN -
-
What is wrong with these reactions explain. CI + NaOCH3 OCH3 + NaCl a) + HBr Br . b) CH,OH + CH;0 OCH3 CI d)
-
How much heat is required to convert 185 g of ice at -8C to water at 8C? cal Additional Materiale
-
If the object-spring system is described by x = (0.325 m) cos (1.05t), find the following. (a) the amplitude, the angular frequency, the frequency, and the period A = .325 W = 1.05 m rad/s f = .167...
-
Two people decide to go skydiving. Jethro has a mass of 7 0 kg , and Joel has a mass of 1 0 0 kg . After they pull their parachutes, which will have a greater terminal velocity? Explain.

Study smarter with the SolutionInn App


