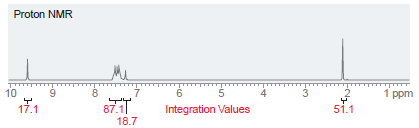
A compound with molecular formula C 10 H 10 O has the following NMR spectrum. Determine the
Question:
Transcribed Image Text:
Proton NMR 10 3 1 ppm 4 87.1| 18.7 Integration Values 17.1 51.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The signal at 96 ppm represents one ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 13 H 10 O produces a strong signal at 1660 cm 1 in its IR spectrum. The 13 C NMR spectrum for this compound is shown below. Identify the structure of this...
-
A compound with molecular formula C 11 H 14 O 2 exhibits the following spectra ( 1 H NMR, 13 C NMR, and IR). Identify the structure of this compound. Proton NMR 11 Chemical Shift (ppm) Carbon NMR...
-
Identify the following compounds. (Relative integrals are given from left to right across the spectrum.) a. The 1H NMR spectrum of a compound with molecular formula C4H10O2 has two singlets with an...
-
A teacher traces a small circle on the palm of a kindergartener's hand to let him know it is time for free play. What type of disability does this child most likely have? A. Traumatic Brain Injury B....
-
"Returns were much higher in the 1970s than in the early 1990s because the interest rates were much higher." True or false?
-
Debt covenants and agency relationships On 1 July 2022, Medical Supplies Ltd borrowed \$15 million to finance an investment in a laboratory for developing and testing surgical supplies. The loan is...
-
For an isothermal reversible change of the system, the work function is (a) \(\left(\frac{\partial A}{\partial V} ight)_{T}=P\) (b) \(\left(\frac{\partial A}{\partial U} ight)_{T}=-P\) (c)...
-
Carbon monoxide at 25C and steam at 150C are fed to a continuous water-gas shift reactor. The product gas, which contains 40.0 mole% H2, 40.0% CO2, and the balance H2O (v), emerges at 500C at a rate...
-
2. Aaron and his friends Britney, Carlos, Diana, Emily, Frank, Gwen, Hugo, Isabela, and Juliana have two choices for weekend activities. They can either go to the neighborhood park or get together in...
-
If the last digit of a weight measurement is equally likely to be any of the digits 0 through 9, (a) What is the probability that the last digit is 0? (b) What is the probability that the last digit...
-
A compound with molecular formula C 5 H 10 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR lle 2.0 1.5 0.5 ppm 4.5 4.0 33.2 3.5 3.0 2.5 1.0...
-
A compound with molecular formula C 4 H 6 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR 25 2.0 2.0 1.5 ppm 3.0 Integration Values 5.0 4.5...
-
You have a controlling interest if: (a) you own more than 20% of a companys stock. (b) you are the president of the company. (c) you use the equity method. (d) you own more than 50% of a companys...
-
5 Complete the table below stating for each account, the account class which it belongs to, its normal account balance and the financial statement where its found (IS-income statement, BS- Balance...
-
Delph Company uses a job-order costing system and has two manufacturing departments-Molding and Fabrication. The company provided the following estimates at the beginning of the year: Machine-hours...
-
The accounting records of NuTronics, Incorporated, include the following information for the year ended December 31: Inventory of materials Inventory of work in process Inventory of finished goods...
-
The following financial statement information is from five separate companies. Company A Company B Company C Company D Company E Beginning of year Assets Liabilities $ 45,000 36,900 $ 35,100 $ 28,800...
-
Your firm is the external auditor for Southpoint Engineering, a publicly listed company with annual sales of $100 million. The company's head office site houses the manufacturing unit, accounting...
-
The graph shows the number of farms in the United States for selected years since 1950. We can use the formula Number of farms = 0.001259x 2 - 5.039x + 5044 to get a good approximation of the number...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
We?ll see that there are two isomeric substances both named 1, 2-dimethylcyclohexane. Explain. -C3 1,2-Dimethylcyclohexane CH
-
Give IUPAC names for the following cycloalkanes: (c) CH2CH2CH3 (b) CH (a) CH CH3 (f) Br (e) C CH-CH (d) CHICH3)2 "CH C(CH3)3 Br
-
Draw structures corresponding to the following IUPAC names: (a) 1, 1-Dimethylcycloocatne (b) 2-Cyclobutylhexane (c) 1, 2-Dichlorocyclopentane (d) 1, 3-Dibromo-5-methylcyclohexane
-
Energy market contains three generation companies and three consumers. The energy bids and offers submitted are presented in the following tables. Plot the supplies and consumer curves. Determine the...
-
During a three - year period, Appliances, Inc., performed electrical heating and plumbing work for Yost Construction worth approximately $ 7 , 0 0 0 . Yost never paid Appliances for any of these...
-
ABC and BCD Joint Corporation is a division of a major corporation. Last year the division had total sales of SAR 8 5 , 7 8 0 , 0 0 0 , net operating income of SAR 8 , 6 9 7 , 5 7 0 , and average...

Study smarter with the SolutionInn App


