Assign a systematic (IUPAC) name for each of the following compounds: a. b. c.
Question:
a.
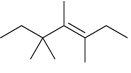
b.
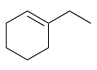
c.
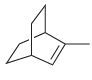
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
a Trans3 4 5 5te...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the IUPAC name for each of the following compounds: (a) CH3(CH2)25CH3 (b) (CH3)2CHCH2(CH2)14CH3 (c) (CH3CH2)3CCH(CH2CH3)2 (d) (e) (f) (g)
-
Give the systematic name for each of the following compounds: a. b. c. d. e. f. CH3CH==CHOCH2CH2CH2CH3 CH CHCH CHCH CH3 CH CH CH C CCHCH CH3 Cl Br BrCH CH CH-CCH CH2CH CH3
-
Give the systematic name for each of the following compounds: a. b. CH2==CHCH2C==CCH2CH3 c. d. e. f. g. CH3CH==CHCH==CHCH==CH2 h. CH3 CH,CH-CCH CH-CH CH CH CH,CH CH CCH CH2CH HOCH,CH C CH CH CH CH...
-
Two spheres of mass 1 kg and 1 . 5 kg hang from the ends of two 1 . 5 meter long threads fixed at the ceiling point. The lighter sphere is pulled to one side so that its thread forms an angle of 6 0...
-
Lebanese american Who is represented in this population? What is the size of this population in the United states? Where are they located? What is the size of this population in Michigan? Where are...
-
Liquid A forms a concave meniscus of radius \(R\) in a capillary tube in which its capillary rise is \(b_{A}\). Liquid \(B\) has twice the surface tension and twice the mass density of liquid...
-
True or False. A limit cycle denotes a steady-state periodic oscillation.
-
1. What clues caused Jane to suspect that fraud was involved? 2. Why is it important for fraud examiners to follow up on even the smallest inconsistencies? 3. In an attempt to identify possible...
-
S You are a relatively recent hire to Hartz & Company, a local manufacturer of plumbing supply products. You have been asked to prepare a condensed statement of cash flows for the months of November...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
A sample of K(s) of mass 2.740 g undergoes combustion in a constant volume calorimeter. The calorimeter constant is 1849 J K 1 , and the measured temperature rise in the inner water bath containing...
-
What are the environmental benefits of resource recovery? Of fluidized-bed combustion?
-
Here are book- and market-value balance sheets of the United Frypan Company: Assume that MM's theory holds except for taxes. There is no growth, and the $40 of debt is expected to be permanent....
-
What are the 3 questions (in order of sequence) to analyze business transactions? Explain.
-
Samantha grossed $ 3200 and $ 704 of that goes to taxes. If the following paycheck noted $ 572 in taxes, what was the gross value?
-
Get a current copy of The Wall Street Journal and find the Dow Jones Country Indexes listing in Section C of the newspaper. Examine the 12-month changes in U.S. dollars for the various national...
-
Kim, a broker appointed with a large life insurance company, is being investigated as part of a larger money laundering investigation. If convicted of money laundering, what does she face?
-
Consider a cutting-stock problem where an unlimited supply of rolls of length 27 is available. Three smaller types of rolls of sizes 7, 9 and 12 are required to be produced to satisfy a demand of 30,...
-
Write the binomial expansion of each expression. (3r + s) 6
-
A company has the following incomplete production budget data for the first quarter: In the previous December, ending inventory was 200 units, which was the minimum required, at 10% of projected...
-
What product would you expect from a Robinson annulations reaction of 2-methyl-1, 3-cyclopentanedione with3-buten-2-one? -CH + H%3DH 2-Methyl-1,3-cyclo- pentanedione 3-Buten-2-one
-
How would you prepare the following compound using a Robinson annulations reaction between a ?-dike tone and ?, ?-unsaturated ketone? Draw the structures of both reactants and the intermediate...
-
What ketone or aldehydes might the following enone have been prepared from by aldolreaction? (b) (a)
-
Select a company you are familiar with and detail one short-term and one longer-term strategy, then discuss how the role of human resource development can provide valuable advice and counsel in the...
-
Sunspot Beverages, Limited, of Fiji uses the weighted-average method of process costing. It makes blended tropical fruit drinks in two stages. Fruit juices are extracted from fresh fruits and then...
-
How does TPM intersect with Lean Manufacturing principles? Discuss the synergies between TPM and Lean practices, such as continuous improvement (Kaizen) and value stream mapping, in reducing waste...

Study smarter with the SolutionInn App


