Cyclopropane (shown below) has only one constitutional isomer. Draw a condensed structure of that isomer. -.
Question:
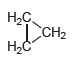
Transcribed Image Text:
Н-С. Т Сн- CH2 H2Č
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
H ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propylene is produced by cracking petroleum and is a very useful precursor in the production of many useful polymers. Propylene has one constitutional isomer. Draw that isomer, and identify its...
-
Two constitutional isomers having molecular formula C4H6O are both symmetrical in structure. In their infrared spectra, neither isomer when in dilute solution in CCl4 (used because it is nonpolar)...
-
Vaccenic acid, a constitutional isomer of oleic acid, has been synthesized through the following reaction sequence: Propose a structure for vaccenic acid and for the intermediates A-E. liq. NaUN KUH,...
-
Your input file contains sentences. A sentence contains words seperated with one space or more spaces. And a sentence ends with a "." Symbol. Write appropriate lex code to create desired outputs....
-
What is the role of arbitragers in futures markets? What impact do they have on futures prices?
-
Construct a Pareto chart of the data given in Exercise 21. Compare the Pareto chart to the pie chart. Which graph is more effective in showing the relative importance of the mistakes made by job...
-
The Lewis-Randall rule is applicable (a) At high pressures (b) At low pressures when the gas mixture behaves as an ideal solution (c) At low temperature (d) At low temperature and high pressure.
-
Adriana Company had a net income of $130,000 and paid cash dividends of $26,000 for 20--. Overman Company had a net loss of $25,000 and distributed a 10% stock dividend with a market value of...
-
Patricia John and Linda Crawford borrowed $13,200 on a 7-month, 5% note from Gem State Bank to open their business, Crane's Coffee House. The money was borrowed on June 1, 2027, and the note matures...
-
Charges -q and +2q in FIGURE P23.39 are located at x = ±a. Determine the electric field at points 1 to 4. Write each field in component form. y 2a +1 2 3 -2a +29 2a -a- -2a +4 FIGURE P23.39
-
Propose a molecular formula for a compound that exhibits the following peaks in its mass spectrum. a) (M) + at m/z = 72, relative height = 38.3% of base peak (M+1) + at m/z = 73, relative height =...
-
Most common amines (RNH 2 ) exhibit pK a values between 35 and 45. R represents the rest of the compound (generally carbon and hydrogen atoms). However, when R is a cyano group, the pK a is found to...
-
The New York Times reported: Several years ago, Google, aware of how many of us were sneezing and coughing, created a fancy equation on its Web site to figure out just how many people had influenza....
-
Competitiveness is defined as how effectively an organization meets the wants and needs of its customers relative to other organizations with similar goods or services. Organizations compete through...
-
In managing conflict in projects, it is prudent to identify the stakeholders and thereafter to analyse them. By referring to a project that you are familiar with, use an appropriate tool to analyse...
-
To be successful and understand if Four Seasons is meeting its goals, you need to develop a marketing plan overview that is specific about what Marketing Goals and Objectives are along with the...
-
You are requested to submit recommendations citing the strategic importance of scheduling for Barloworld Logistics and to critically assess the strategies that you would consider in influencing...
-
1. When we are talking about Industry 4.0, society understands that industry more integrated and supply chain will more effectively to help industry 4.0. Please find an example of industry 4.0 in...
-
Factor the trinomial, if possible. 9x 2 + 4x - 2
-
Question 2 For an n x n matrix A = form) via (aij)
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked by an asterisk disappears when D2O is added to thesample. TMS 10 8. 0 ppm Chemical shift (8)...
-
The reduction of carbonyl compounds by reaction with hydride reagents (H: ? ) and the Grignard addition by reaction with organo magnesium halides (R: ? + MgBr) are examples of nucleophilic carbonyl...
-
What is Preferred Stock? Discuss the characteristics of Preferred Stock over Common Stock. MINIMUM ONE PAGE REQUIREMENT. 2. What are Stock Options? Discuss the TWO primary reasons why corporations...
-
If your organization was given $10,000 for digital marketing for a quarter (3 months), how would you spend it?
-
For the following function, list the intervals where f(x) is concave up and where it is concave down. Find any points of inflection. Do NOT use a graph to answer the question. f(x)=3x+2x-12x+5

Study smarter with the SolutionInn App


