Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each
Question:
a.
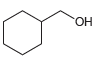
b.
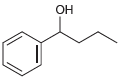
c.
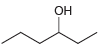
d.
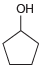
Transcribed Image Text:
Но. Он ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a b c...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how each of the following alcohols can be made from a Grignard reagent and a carbonyl compound: a. b. 0 0
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
The RRR Company has a target current ratio of 2.4. Presently, the current ratio is 3.3 based on current assets of $6,567,000. If RRR expands its inventory using short- term liabilities (maturities...
-
On January 10, VW agrees to import auto parts worth $7 million from the US. The parts will be delivered on March 4 and are payable immediately in dollars. VW decides to hedge its dollar exposure by...
-
What growth opportunities do you envision for Ita Unibanco and what is the best organizational approach to develop and implement these growth strategies?
-
Discuss the reasons why almost all colonies had achieved independence by the end of the 1960s.
-
Deluxe Ezra Company purchases equipment on January 1, Year 1, at a cost of $469,000. The asset is expected to have a service life of 12 years and a salvage value of $40,000. Instructions (a) Compute...
-
Assume that a Temperature Sensor is interfaced to 8 0 5 1 microcontroller to sense the temperature of the room. Design a block diagram and a circuit diagram to explain the interfacing of the...
-
Determine the mass of an ethane gas stored in a 25 ft3 tank at 250 F, 440 lbf/in.2 using the compressibility chart. Estimate the error (%) if the ideal gas model is used.
-
Starting with 1-butanol, show the reagents you would use to prepare each of the following compounds. a. b. c. d. e. H. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Define binary tree. Explain different types of binary trees.
-
duction Fair value accounting and historical cost method are important measurements that equally have a place in the industry to be followed. Experts have examined whether fair value accounting is...
-
You are a new auditor at Assure Pro Accounting Firm. You have been assigned to work with your first client, Ventura Medical Group. Before beginning your work with this client, your manager has asked...
-
Sam works for Axis Entertainment, Inc. As part of his employment he is given a laptop computer. Sam uses the laptop for work and occasionally uses the laptop for PERSONAL USE. On the computer he has...
-
Rosebush v. Oakland: On what basis does the court conclude that adults have a right to refuse life-sustaining treatment? Why do parents have a right to withdraw life sustaining treatment from their...
-
. Discuss the evidence of gang activity in the community. Do you have a gang problem in your community? If so, do you see your community responding to this threat?
-
Identify the type of graph that each equation has, without actually graphing. x 2 + y 2 = 144
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
Isoleucine and threonine are the only two amino acids with two chirality centers. Assign R or S configuration to the methyl-bearing carbon atom ofisoleucine.
-
Is the following structure a D amino acid or an L amino acid? Identifyit.
-
Give the sequence of the following tetrapeptide:
-
Image transcription text 1. A cylindrical specimen of cold-worked copper (see Fig. below) has experienced a ductility of 25%EL. If its cold worked radius is 10 mm, what was its radius before...
-
Question 2 As a project manager, you need to synthesize information about a problem to stakeholders. What should you do first in your synthesizing process? Describe Briefly.
-
Please help me as much as you can!! I will take all your effort to solve this problem and will give you a good rate!!! Please show all the calculations in detail! Also, please do not copied and...

Study smarter with the SolutionInn App


