Predict the product(s) for each of the following reactions: (a) (b) (c) OH HNO, H,SO, OMe Br
Question:
(a)
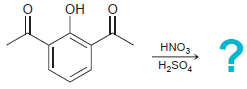
(b)
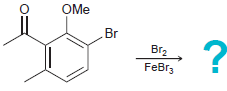
(c)
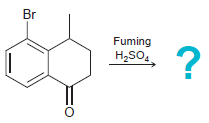
Transcribed Image Text:
OH HNO, H,SO, OMe Br Br2 FeBr3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
a b c...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Find the area of the equilateral triangle of a side of 5 using the Babylonian way. A. 10;1,20 . 10:56,15 . 10:26,15 D. 10:25 A B C
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) (b) (c) (d) ...
-
For Example, 10.3 with = 0, verify that the stresses from equation (10.5.18) reduce to those previously given in Eq. (8.4.69). Data from example 10.3 Equation 10.5.18 Equation 8.4.69 Consider next...
-
Describe the common methods of establishing an advertising budget. Which method is most often used? Which technique is most often recommended? Why? Identify four basic methods for preparing an...
-
Has Jill established the attorney-client relationship or has that already been established?
-
Use a scatter plot to display the data shown in the table at the left. The data represent the numbers of coaching hours and the hourly fees (in dollars) of 12 cricket coaches. Organize the data using...
-
I know that its the thing to do, insisted Pamela Kincaid, vice president of finance for Colgate Manufacturing. If we are going to be competitive, we need to build this completely automated plant. Im...
-
Jane Smith is a client that has come to you seeking start-up advice on designing an accounting system to accumulate the costs associated with her new home construction business, a business in which...
-
What is a decision statement? How does the focus on an irrelevant decision affect the research process?
-
For each compound below, identify which position(s) is/are most likely to undergo an electrophilic aromatic substitution reaction. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH3 NO2 O,N. CH3 O,N NO2
-
Calculate K P at 600.K for the reaction N 2 O 4 (l) 2NO 2 (g) assuming that H o R is constant over the interval 298 725 K.
-
Does a car speedometer measure speed, velocity, or both?
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. Acts of an employee while performing duties of employment are considered the acts of the...
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. An employee who performs only mechanical acts under the employers direction and is...
-
A principal whose identity is not known to a third party who knows he or she is dealing with an agent. a. agent b. agency by necessity c. agency by ratifi cation d. apparent a uthority e. attorney in...
-
Would a projects NPV for a typical firm be higher or lower if the firm used accelerated rather than straight-line depreciation? Explain.
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. In a contract between an agent and a third party, the agent can be held responsible to...
-
Sketch the graph of an appropriate function and then use the graph to solve each equation or inequality. First determine whether the expression is an equation or an inequality. When appropriate, use...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Show the elimination reactions that account for each of the following fragments in the CI mass spectrum of di-sec-butyl ether (Fig. 12.17b). (Fig. 12.17b) m/z =115 100 75 base peak131 80 73 60 101 40...
-
Show the elimination reactions that account for each of the following fragments in the CI mass spectrum of di-sec-butyl ether (Fig. 12.17b). (Fig. 12.17b) m/z =115 100 75 base peak131 80 73 60 101 40...
-
List two factors that determine the intensity of an infrared absorption.
-
The initial margin on a GBP futures contract is $2035 and maintenance is $1850. You buy one contract (62,500 Pounds) at $1.3100 and place $2035 in your account. The price of your contract drops to...
-
1.Define a).Market research transnational firm b).Target market c).Maslow's hierarchy of needs d).ethnocentric polycentric Geocentric Explain Please create a response to the question below that is...
-
Describe one product development recommendation and one market development recommendation for Tesla. Explain why this idea should be adopted by the Tesla EV company in the New Zealand market.

Study smarter with the SolutionInn App


