For each of the compounds below determine whether any of the nitrogen atoms bear a formal charge:
Question:
a.
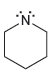
b.
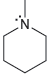
c.
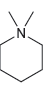
d.
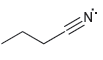
Transcribed Image Text:
N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a b No...View the full answer

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the compounds below determine whether any of the oxygen atoms bear a formal charge: a. b. c. d. ::
-
For each of the compounds below, locate the pattern we just learned (lone pair next to a Ï bond) and draw the appropriate resonance structure: a. b. c. d. e. f. g. h. NH2
-
For each of the compounds below, locate the lone pair adjacent to a positive charge and draw the resonance structure: a. b. c. N.
-
Refer to E 29 and respond to the following requirements. Data in E 2-9 Prepare the necessary adjusting entries on December 31, 2024, for the Microchip Company for each of the following situations....
-
On January 1, 2010, ABC Co. had inventory of 200 units @ 18.00 a unit. It purchased 400 more units @ $20.00 a piece on Feb. 19th. On May 12th, it bought 300 more units @ $22.00 each. On Oct. 3rd, it...
-
Nespresso, a division of Nestles SA, pioneered the development of the single serving coffee ma chine in 1986. By 2009, Nespresso had achieved sales of over $2.6 billion with double-digit growth...
-
P(x > 185) The random variable x is normally distributed with mean = 174 and standard deviation = 20. Find the indicated probability.
-
Owl, Inc., was owned entirely by Jeri Bell and Jerry Gore, each owning 620,000 of the 1,240,000 shares of common stock outstanding. On January 1, 2015, Owl established an employee stock ownership...
-
For a "normal" $300m venture capital partnership: 1.How many portfolio companies would you expect? 2.How many of the portfolio companies do you expect to fail at a 100% loss to the VC partnership?...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Atenolol and enalapril are drugs used in the treatment of heart disease. Both of these drugs lower blood pressure (albeit in different ways) and reduce the risk of heart attack. Using the following...
-
Draw all lone pairs on each of the oxygen atoms in the compounds below. Before doing this, review in the following table, and then come back to these problems. Try to identify all lone pairs without...
-
Refer to the information in E85. Required: Indicate the effects (accounts, amounts, and + or ) of the following on the accounting equation. Date Assets = Liabilities + Stockholders Equity 1. The...
-
The ___________ method measures the proportion of the present value of dollars returned to dollars invested. a. IRR b. NPV c. Profitability index d. Risk-adjusted payback e. None of these
-
The IRR method allows a ranking of competing projects. a. True b. False
-
What does a low ratio of average reserves per well indicate?
-
How is a dry appraisal well accounted for? a. Charged to dry hole expense b. Remains capitalized so long as a subsequent appraisal well is either planned or underway c. Capitalized if other...
-
The IRR method assumes which of the following? a. All future cash inflows will be reinvested at the same rate of return as the IRR. b. None of the future cash inflows will be reinvested at the same...
-
What is MySQL?
-
Where are the olfactory sensory neurons, and why is that site poorly suited for their job?
-
Starting with an appropriate alkyl halide and base, outline syntheses that would yield each of the following alkenes as the major (or only) product: (a) (b) (c) (d) (e)
-
Predict the more stable alkene of each pair: (a) 2-methyl-2-pentene or 2,3-dimethyl- 2-butene; (b) Cis-3-hexene or trans-3-hexene; (c) 1-hexene or cis-3-hexene; (d) Trans-2- hexene or...
-
Arrange the following alcohols in order of their reactivity toward acid-catalyzed dehydration (with the most reactive first): 1-Pentanol 2-Methyl-2-butanol 3-Methyl-2-butanol
-
Melissa Cutt is thinking about buying some shares of EZLawn Equipment, at $36.44 per share. She expects the price of the stock to rise to $43.62 over the next 3 years. During that time she also...
-
At the beginning of the week, Taylan's restaurant had a beginning inventory of food and beverages totaling $4,000. During the week, additional food and beverages purchases totaled $3,000. At the end...
-
1. Identify the auditor's role in the internal and external auditing processes. 2. Identify one law, regulation, and statue affecting healthcare

Study smarter with the SolutionInn App


