For each of the compounds below, locate the pattern we just learned (lone pair next to a
Question:
a.
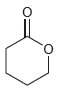
b.
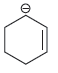
c.
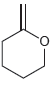
d.
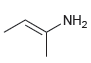
e.
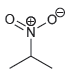
f.
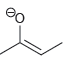
g.
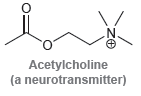
h.
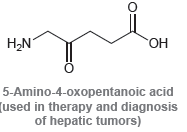
Transcribed Image Text:
NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the compounds below determine whether any of the nitrogen atoms bear a formal charge: a. b. c. d. N.
-
For each of the compounds below determine whether any of the oxygen atoms bear a formal charge: a. b. c. d. ::
-
For each of the compounds below, locate the lone pair adjacent to a positive charge and draw the resonance structure: a. b. c. N.
-
Mixing together solutions of acetic acid and sodium hydroxide can make a buffered solution. Explain. How does the amount of each solution added change the effectiveness of the buffer?
-
Ozark Corporation reported net income of $100,000 for 20X5. The income statement revealed sales of $1,000,000; gross profit of $520,000; selling and administrative costs of $340,000; interest expense...
-
Concern over an aging product line led the Wall Street Journal to publish a chart with a cover-page article on future profits of drug-maker Pfzer.7 Is it a bar chart in the sense of this chapter or a...
-
To the left of z = 0.15 and to the right of z = 0.15 Find the indicated area under the standard normal curve. If convenient, use technology to find the area.
-
Wolsey Industries Inc. expects to maintain the same inventories at the end of 20Y3 as at the beginning of the year. The total of all production costs for the year is therefore assumed to be equal to...
-
Read the following article, A Day in the Life of a PreK Teacher . Share your thoughts about how this way of life each day will suit you well in your strengths as a teacher and where you anticipate...
-
1.3 Determine the force P necessary for the equilibrium of a steel bar shown in Fig. 1.69. The diameters of the first, middle and the last segments of the bar are 30 mm, 25 mm and 30 mm respectively....
-
An ideal gas undergoes a single-stage expansion against a constant external pressure P external = P f at constant temperature from T,P i ,V i , to T,P f ,V f . a. What is the largest mass m that can...
-
Draw the resonance structure(s) for each of the compounds below: a. b. c. d.
-
Sara Lehn, chief financial officer of Merit Enterprise Corp., was reviewing her presentation one last time before her upcoming meeting with the board of directors. Merits business had been brisk for...
-
Look at this partial class definition, and then follow the subsequent instructions: a. Write a constructor for this class. The constructor should accept an argument for each of the fields. b. Write...
-
One of the GLOBE Project teams nine cultural variables is future orientation, which refers to the level of importance a society attaches to future-oriented behaviors, such as planning and investing...
-
On Wednesday, November 21, the WeChat account of the official Peoples Daily issued a statement believed to be from the Ministry of Culture and Tourism; the Dolce & Gabbana (D&G) fashion show in...
-
Refer to the facts in Tax Form/Return Preparation Problem C:9-58. Now assume the company is an S corporation rather than a partnership. Additional facts are as follows: Drs. Bailey and Firth formed...
-
In pairs, imagine that you are buyers from one country and sellers from another. Your colleague says that it is not necessary to spend time and effort studying the selling-country culture before the...
-
How do you create a new E-R diagram in the MySQL Workbench?
-
When is the indirect pattern appropriate, and what are the benefits of using it?
-
Write structures for the major organic products from the following reactions. Show stereoisomers where applicable. (a) (b) (c) (d) (e) H20, H.So4 dr (1) BH THF (2) NaOH, H20, Br (1) 03 (2) Meps
-
Give the structure of the products that you would expect from the reaction of 1-butyne with: (a) One molar equivalent of Br2 (b) One molar equivalent of HBr (c) Two molar equivalents of HBr (d) H2...
-
Provide mechanistic explanations for the following observations: (a) (b) HCI CI CI Cl CI HCI
-
A consultant advises that a fund pays out $100,000 at the end of 5 years. Provided $12,000 is invested in the fund at the start of each year, beginning immediately and continuing up to and including...
-
Durco Automotive needs a $2 million balance in its contingency fund 3 years from now. The chief financial officer (CFO) wants to know how much to deposit now into Durco's high-yield investment...
-
3. Create a chart for gross margin less R&D and capital costs calculations. a. Calculate gross margin less R&D and capital costs for options A and B. b. Graph total gross margin less R&D and capital...

Study smarter with the SolutionInn App


