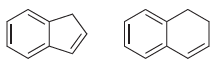
Identify which of the following compounds is more acidic and explain your choice.
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
The first compound is more acidic beca...View the full answer

Answered By

Amar Kumar Behera
I am an expert in science and technology. I provide dedicated guidance and help in understanding key concepts in various fields such as mechanical engineering, industrial engineering, electronics, computer science, physics and maths. I will help you clarify your doubts and explain ideas and concepts that are otherwise difficult to follow. I also provide proof reading services. I hold a number of degrees in engineering from top 10 universities of the US and Europe.
My experience spans 20 years in academia and industry. I have worked for top blue chip companies.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify which of the following compounds is most activated toward electrophilic aromatic substitution. Which compound is least activated? Br NO2 NO2 OMe .
-
Identify which of the following compounds is more acidic. Explain your choice. C=C H--
-
Identify which of the following compounds is expected to have a larger λ max :
-
Acceleration is the rate of change of velocity with time. Is the acceleration vector always aligned with the velocity vector? Explain.
-
Clay Travel, Inc. routinely funds its checking account to cover all checks when written. A thorough analysis of its checking account discloses that the firm could maintain an average account balance...
-
(L05) Roger, a single taxpayer, owns and operates a video game store. He does not have any employees and reported qualified business income of \(\( \$ \) 125,000\) through a sole proprietorship in...
-
Marion Mosley created the Jenny Justice Trust in 2005 with First Bank named as trustee. For 20 years, the trust is to pay out all its income semiannually to the beneficiary, Jenny Justice. At the end...
-
Rossman Instruments, Inc., is considering leasing new state-of-the-art machinery at an annual cost of $900,000. The new machinery has a fouryear expected life. It will replace existing machinery...
-
Consider the following article: https://www.employment-studies.co.uk/system/files/resources/files/mp116.pdf Question: 1. What kind of managerial/leadership style would be most conducive to successful...
-
Derivation of the equation of change for the Reynolds stresses. At the end of S5.2 it was pointed out that there is an equation of change for the Reynolds stresses. This can be derived by (a)...
-
Predict which compound will react more readily in an S N 1 process, and explain your choice. Br -Br
-
For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a. b. c. d. e. f. g. h. :N-H
-
When Turner Company adopted its defined benefit pension plan on January 1, 2019, it awarded retroactive benefits to its employees. These retroactive benefits resulted in a prior service cost of...
-
Builtrite is considering the purchase of a new five-year machine worth $90,000. It will cost another $20,000 to install the machine and Builtrite will need to keep an extra $7,000 in inventory on...
-
Johnny's Milkshakes is producing an income statement for last year. What category of expense should their utility costs (electricity) go under?
-
Natasha earns revenue based on the value of the investment assets that she manages for her clients. What kind of remuneration structure does Natasha use?
-
In the "big history" view, if we were to put all of past time since the beginning of the universe on a 12-month calendar, then recorded human history would begin in the last second of the evening on...
-
While performing a routine audit, an organization discovers it has been overpaid for a claim. What is the next step?
-
Perform each operation, if possible. -2 1 -1 8 2.
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
Write the enantiomeric forms of bromochlorofluoromethane and assign each enantiomer its correct (R) or (S) designation.
-
The relative rates of ethanolysis of four primary alkyl halides are as follows: CH3CH2Br, 1.0; CH3CH2CH2Br, 0.28; (CH3)2CHCH2Br, 0.030; (CH3)3CCH2Br, 0.00000042. (a) Is each of these reactions likely...
-
Classify the following solvents as being protic or aprotic: formic acid, HCO2H; acetone, CH3COCH3; acetonitrile, CH3CN; formamide, HCONH2; sulfur dioxide, SO2; ammonia, NH3; trimethylamine, N(CH3)3;...
-
Install on ubuntu , please provide a screenshot for each step 1)How to install base64 on ubuntu 2)What kind of analysis is performed by Cuckoo? How to install Cuckoo on ubuntu?
-
How do I access my computer files on Windows from Ubuntu? Google is not helping. I tried searching for it in Files on Ubuntu but all that were shown were Ubuntu files, not my computer files.
-
You throw a nerf basketball of mass 0.002 kg into the air. Att = 0, the ball leaves your hand. Att = 0.10 s, the net force on the b (-0.0203,-0.0248, 0) N, its velocity is (1.87, 0.84, 0) m/s, and...

Study smarter with the SolutionInn App


