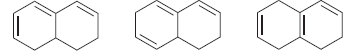
Identify which of the following compounds is expected to have a larger λ max :
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify which of the following compounds is expected to be a stronger base. Justify your choice. N. N.
-
Identify which of the following compounds is expected to have the larger heat of combustion:
-
Identify which of the following compounds is more acidic and explain your choice.
-
Guidance Residential in Reston, Virginia, offers a Shari'ah-compliant housing finance product for Muslims and others who do not believe in collecting or paying interest. Under their Declining Balance...
-
An unconfined compression test is performed on a dense silt. Previous drained triaxial tests on similar samples of the silt gave ' = 32o. If the unconfined compressive strength was 420 kPa, estimate...
-
Refer to the Museum Management and Curatorship (June 2010) worldwide survey of 30 leading museums of contemporary art, Exercise. Recall that each museum manager was asked to provide the performance...
-
A SIF uses a single, pneumatic, final element assembly consisting of a 3/2 pilot-operated poppet solenoid valve, a spring-return pneumatic piston actuator, and a floating ball valve. The application...
-
A liquid mixture consisting of 100 kmol of 60 mol% benzene, 25 mol% toluene, and 15 mol% o-xylene is flashed at 1 atm and 100C. (a) Compute the amounts of liquid and vapor products and their...
-
Would you be able to recreate the planning and strategic management process that Mr. Nadella used? What would Microsoft's planning flowchart look like based on the goal he proposed? Did Mr. Nadella...
-
The Bombay Beverage Company has heavily advertised its social responsibility to the environment and community. In its advertisements, the company touts how its recycling efforts have reduced its...
-
The Cordell Coffee Company is evaluating the within-plant distribution system for its new roasting, grinding, and packing plant. The two alternatives are (1) a conveyor system with a high initial...
-
The after-tax cash flows for two mutually exclusive projects have been estimated, and the following information has been provided: The companys required rate of return is 14 percent, and it can get...
-
An electron of kinetic energy 45 keV moves in a circular orbit perpendicular to a magnetic field of 0.325 T. (a) Find the radius of the orbit. (b) Find the frequency and period of the motion.
-
We would not need to economize if _____. a) the government printed more money b) there was no scarcity c) there was less output of goods and services d) everyone received a big pay increase
-
Charging flood victims $30 for a case of water or $10 for a gallon of gas doesnt sit right. And a majority of states, including Texas, have laws against price gouging. The state attorney general has...
-
A pharmacist received a bottle of cephalosporin capsules. Unknown to the pharmacist, the capsules also contained small amounts of penicillin. The pharmacist dispensed the capsules to a patient who is...
-
The FDA issued a final compliance guidance related to pharmacy compounding. In the guidance, the FDA clarified which activities compounding pharmacies could lawfully engage and which activities the...
-
Explain how each of the following actions will affect the level of planned investment spending and unplanned inventory investment. Assume the economy is initially in incomeexpenditure equilibrium. a....
-
Go to the IRS website (www.irs.gov) and print out a copy of the most recent Schedule F of Form 1040.
-
The Strahler Stream Order System ranks streams based on the number of tributaries that have merged. It is a top-down system where rivers of the first order are the headwaters (aka outermost...
-
The following structure represents a tetrahedral alkoxide-ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the...
-
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions?...
-
Give IUPAC names for the following compounds: o (c) CHH2H2 CH2CH3 (a) (b) CH3CH2CHCHI NH2 (d) (e) (f) CH3CHCH,NHCH3 CH-CH-C H CH Br (h) (g) SCH(CH3)2
-
The highest WACC is 11%, the baseline WACC is 7.4% and lowest WACCS is 2.8%. To derive a WACC that better accounts for extreme scenarios, you decide to assign the following probability weights: High-...
-
Give 2 examples of the ff: Statutory law Regulatory law Common law Include the title of each example and explain why you chose these examples ( 1 to 2 sentences ) . Note: Write your reference ( s )
-
The Adams family includes a financially well-informed couple, both aged 36, and two children aged 4 and 6. The family is financially sound but suffered badly during the tech meltdown in 2000. The...

Study smarter with the SolutionInn App


