Predict the major product(s) for each of the following reactions: a. b. c. d. Br2 Br2
Question:
a.
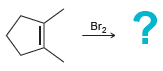
b.
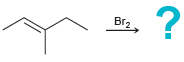
c.
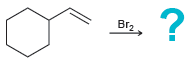
d.
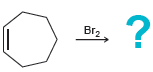
Transcribed Image Text:
Br2 Br2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a b ...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
.How dependent do you think the success of zero-based budgeting would be on the types of attribution models used? How might you increase the odds of success in this budgeting approach? .Advertising,...
-
What authorizations does the AR Supervisor not have for Account Types? Why do you think the AR Supervisor would be restricted from these authorizations for this accounting document?
-
Write a static method max3() that takes three int arguments and returns the value of the largest one. Add an overloaded function that does the same thing with three double values.
-
Find the exact solution of the nonlinear pendulum equation \[\ddot{\theta}+\omega_{0}^{2}\left(\theta-\frac{\theta^{3}}{6} ight)=0\] with \(\dot{\theta}=0\) when \(\theta=\theta_{0}\), where...
-
Boulware Products, Inc. produces printers for wholesale distributors. It has just completed packaging an order from Shawl Company for 450 printers. Before the order is shipped, the controller wants...
-
construct a timeline of significant moments of intersection between Indigenous Peoples and the Australian Constitution. 2. what are some of the critical omissions in the Australian Constitution? 3....
-
Let Ai be the set of all nonempty bit strings (that is, bit strings of length at least one) of length not exceeding i. Find (a) (b) UA
-
Compound X has molecular formula C 5 H 10 . In the presence of a metal catalyst, compound X reacts with one equivalent of molecular hydrogen to yield 2-methylbutane. a. Suggest three possible...
-
Predict the major product(s) that are expected when each of the following alkenes is treated with Br 2 /H 2 O: a. b. c. d.
-
The Valley United Soccer Club has boys and girls travel soccer teams at all age levels up to 18 years. The club has been successful and grown in popularity over the years; however, an obstacle to its...
-
You are also told that the prevailing risk free rate is 4%, the market risk premium is 5%, and MM has a of 0.8. With all this as a backdrop, show that MM's stock is correctly priced at $25/share.
-
Shown below is a recent income statement for B-D Electric. B-D ELECTRIC Income Statement For the Year Ended January 31, Year 2 Net sales $ 7,500,000 Less: Cost of goods sold 4,100,000 Gross profit $...
-
In the current year, Aveeno reported net income of $57,680, which was a 12% increase over prior year net income. Compute prior year net income. Prior year net income
-
What is the time complexity of the following algorithm: m=a i=1 while (i
-
Bike-O-Rama produces two bike models: Voltage and EasyRider. Departmental overhead data follow. Department Fabricating Assembly Required: Budgeted Cost $ 135,800 144,000 Allocation Base Machine hours...
-
Evaluate the series. (5 (5i + 2) i=3
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Which of the following compounds would you expect to have a ? ? ?* UV absorption in the 200 to 400 nm range? (a) (b) (c) (CH3)2C=C=Do CH2 A ketene Pyridine
-
Predict the products of the following Diels?Alder reactions; (b) (a)
-
Show the structure, including stereochemistry, of the product from the following Diels?Alder reaction: COCH3 C
-
Suppose you were interested in studying the quality of conditions within a prison. What indicators would you measure to give the clearest picture of the realities of prison life? Cite the below...
-
On March 31, 2023, Panda Co. assessed its assets for impairment as part of its year-end procedures. It was found that equipment had a recoverable value of $15,000, a remaining useful life of three...
-
Petty's comparative balance sheets at December 31, 2020, and December 31, 2019, report the following (in millions). (Click the icon to view the comparative balance sheets.) Requirements Below are...

Study smarter with the SolutionInn App


