Predict the major product(s) that are expected when each of the following alkenes is treated with Br
Question:
a.
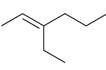
b.
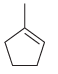
c.
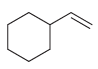
d.
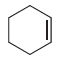
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products that are expected when each of the following alkenes is treated with a peroxy acid (such as MCPBA) followed by aqueous acid: a. b. c. d. e. f.
-
Predict the products that are expected when each of the following alkenes is treated with ozone followed by DMS: a. b. c. d. e. f.
-
Draw the major product(s) that are expected when each of the following amines is treated with excess methyl iodide and then heated in the presence of aqueous silver oxide. (a) (b) NH2 NH2
-
What is the formula to find total dividend and payout ratio? This is the information I have: the amount of shares the company holds and the last dividend paid. Lastly, will there be enough cash to...
-
The control environment reflects the company's attitude, awareness, and actions of management and the board concerning the importance of control and how it is used. This is very relevant to an...
-
Define politics and explain how politics differs from power.
-
A simple pendulum of length \(0.8 \mathrm{~m}\) is released from the initial position of \(80^{\circ}\) from the vertical. How long does it take to reach the position \(\theta=0^{\circ}\) ?
-
1. Identify the ethical culture problem at Texaco in the mid-1990s. 2. Based on the facts in the case and what you have learned in this chapter, evaluate the culture change effort that is under way....
-
What is the role that honor plays in this society in terms of blameworthiness, or in terms of how forgivable/unforgiveable certain behaviors are? Whose honor are we talking about here? Whose emotions...
-
An outpatient clinic has kept a record of walk- in patients during the past year. The table below shows the expected number of walk- ins by day of the week: The clinic has a staff of five physicians,...
-
Predict the major product(s) for each of the following reactions: a. b. c. d. Br2 Br2
-
Muscalure is the sex pheromone of the common housefly and has the molecular formula C 23 H 46 . When treated with O 3 followed by DMS, the following two compounds are produced. Draw two possible...
-
The percentage of the worlds population living in extreme poverty dropped significantly from 42.5% in 1981 to 9.2% in 2017. If the percentage declined linearly during that time, estimate the...
-
Assume that you are the database security analyst for the TECHTEC LLC. The employees are mostly working in shift basis. When 50% of the employees are working from home due to the covid-19 pandemic by...
-
3. Suppose that you are interested in choosing a set of investments {1,,7} using 0 1 variables. Model the following constraints using only linear equations (obs: operators such as or or if can not be...
-
Typical consumer's basket: 20 pizzas, 10 compact discs. Find the CPI and Inflation rate below data if the base year is 2002. prices: pizza CDs 2002 $10 $15 2003 $11 $15 2004 $12 $16 2005 $13 $15
-
Covington Corporation's comparative balance sheet for current assets and liabilities was as follows: Dec. 31, 20Y2 Dec. 31, 20Y1 Accounts receivable $16,500 $15,800 Inventory 79,000 79,700 Accounts...
-
5 9 Record the journal entries for the June 7 session date. Payment Cheque #488 Dated June 5, 2021 To Red Deer Promotions, $2 940 in payment of invoice #RDP-8668. Receipt #120 Dated June 5, 2021 From...
-
Evaluate the series. (6-31) i=2
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
How can you account for the fact that cis-1, 3-pentadicne is much less reactive than trans-1, 3-pentadiene in the DielsAlder reaction?
-
Would you expect a conjugated diyne such as 1, 3-butadlyne to undergo DielsAlder reaction with a dienophile explain.
-
Reaction of isoprene (2-methyl-1, 3-hutadiene) with ethyl propenoate gives a mixture of two Diels?Alder adducts. Show the structure of each, and explain why a mixture is formed. CO2CH2CH3
-
Suppose the correlation between the stock euro returns of Siemens and the USD/EUR exchange rate is 0.2. The standard deviation of the USD/EUR is 10% and the standard deviation of Siemens's stock euro...
-
list and describe the three key client-related factors that the advisor is required to consider when developing a "suitable" investment portfolio for their client. Please cite resources used
-
Year 1 2 3 Amount ($) 2000 3000 4000 An investment made today will pays you the above cash flows at the end of each year. If your required rate of return is 5% annual interest, how much will you pay...

Study smarter with the SolutionInn App


