Which of the following compounds would you expect to have a ? ? ?* UV absorption in
Question:
Which of the following compounds would you expect to have a ? ? ?* UV absorption in the 200 to 400 nm range?
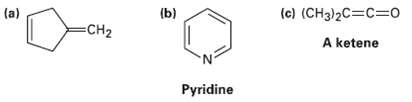
Transcribed Image Text:
(a) (b) (c) (CH3)2C=C=Do CH2 A ketene Pyridine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Only compounds having alternatin...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following compounds would you expect to have a dipole moment? If the molecule has a dipole moment, specify its direction. (b) H2O (c) CH4 (d) CH3Cl (e) CH2O (f) HCN
-
Which of the following compounds would you expect to be the most generally reactive, and why? DO
-
Which of the following compounds would you expect to be most acidic? Justify your choice.
-
If two krypton atoms are held together by a stabilization energy of 1.005 kJ/mol at what temperature would you expect these atoms to transition from bound to unbound? Show your thought process for...
-
What are the goals of management, unions, and society in this situation?
-
For the following Two-loop Electric Circuit System, R1 Vin(t) R www DO in(t) :C iz(t) the mathematical models applied for the voltages are given in time-domain as: Vr(t) = Ri(t) di(t) V (t) = L dt...
-
In a process for the manufacture of \(\mathrm{C}\) from \(\mathrm{A}\) and \(\mathrm{B}\), streams of \(\mathrm{A}\) and \(\mathrm{B}\) are fed to a CSTR, where they undergo reversible reaction to...
-
Consider a two-period model with two firms, A and B. In the first period, they simultaneously choose one of two actions, Enter or do not enter. Entry requires the expenditure of a fixed entry cost of...
-
John wants to be able to withdraw $10,426 every year for 14 years from his retirement fund 1 year after he retires. How much he must accumulate in his retirement account if the interest rate is 4%...
-
CSC20 Project 4 For this project our aim is to create a phone book with contact details. Individual contact information is added to the contact class. Then these contacts are added to the Phone books...
-
Would you expect allene, H2C = C = CH2, to show a UV absorption in the 200 to 400 urn range? Explain.
-
Predict the products of the following Diels?Alder reactions; (b) (a)
-
A random sample was taken from the Financial Post list of top 500 companies for 1998. Of 26 food- and beverage-related industries in the sample, 12 had at least 10% foreign ownership. Of 29 wholesale...
-
Cathy, an Aboriginal woman, is being supported by staff after the loss of her home and her son in a bushfire. Explain how staff should communicate with Cathy to respond to her needs in relation to...
-
Using a four-month moving average on the data given in the spreadsheet, determine one-step-ahead and two-step-ahead forecasts for July through December 2016. Compare their performance. (Hint: In the...
-
The principal at your school decides to retire, and several well-qualified teachers on the staff apply for the position. Mr. England has been teaching for four years and has his administrator's...
-
Several sectors in the global economy have been hit especially hard due to the COVID-19 pandemic. A couple of the hardest-hit sectors include automotive, healthcare services, pharma, hospitality, and...
-
Define values, heroes, rituals and symbols. How do these elements influence consumer behaviour in an international context?
-
Solve the IVP. Do not use Laplace transformation. \(\dot{y}=(1-y) \sin t, y\left(\frac{1}{2} \pi ight)=2\)
-
Two mutually exclusive investment alternatives are being considered. Alternative A requires an initial investment of $20,000 in a machine. Annual operating and maintenance costs are anticipated to be...
-
Which of these aqueous solutions has the highest boiling point? a) 1.25 M C 6 H 12 O 6 b) 1.25 M KNO 3 c) 1.25 M Ca(NO 3 ) 2 d) None of the above (they all have the same boiling point)
-
A graduate student tried to make o-fluorophenylmagnesium bromide by adding magnesium to an ether solution of o-fluorobromobenzene. After obtaining puzzling results with this reaction, she repeated...
-
A common illicit synthesis of methamphetamine involves an interesting variation of the Birch reduction. A solution of ephedrine in alcohol is added to liquid ammonia, followed by several pieces of...
-
Show how you would use a Suzuki reaction to synthesize the following biaryl compound. As starting materials you may use the two indicated compounds, plus any additional reagents you need. Make OCH3...
-
The increasing volatility in the global economy has caused investors to seek out safer investments alternatives. Risk is inevitable in all investments. A capital budget is used to help investors plan...
-
You invest 50% of your financial assets in the Vanguard Total Stock Market ETF (VTI) and 50% in the Vanguard Total Bond Market ETF (BND). VTI has an expected return of 8% and a standard deviation of...
-
You have found the car you would like to purchase. The negotiated price = $57,500 and you are taking out a loan with a 7.5% APR compounded monthly for 6 years. What are your monthly payments? Car...

Study smarter with the SolutionInn App


