Propose a structure that is consistent with each of the following 1 H NMR spectra. In each
Question:
(a)
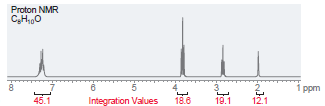
(b)
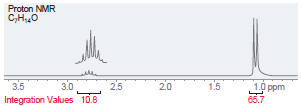
(c)
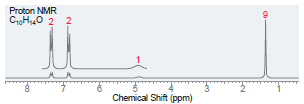
(d)
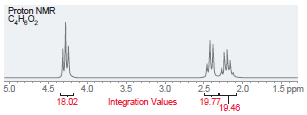
(e)
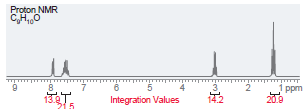
(f)
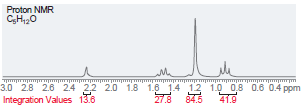
Transcribed Image Text:
Proton NMR C,H,,0 i ppm Integration Values 18.8 12.1 19.1 45.1 Foo Proton NMR C,H0 3.5 Integration Values 10.8 1.0 ppm 85.7 2.5 2.0 1.5 3.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a ...View the full answer

Answered By

Mehwish Aziz
What I have learnt in my 8 years experience of tutoring is that you really need to have a friendly relationship with your students so they can come to you with their queries without any hesitation. I am quite hardworking and I have strong work ethics. Since I had never been one of those who always top in the class and always get A* no matter what, I can understand the fear of failure and can relate with my students at so many levels. I had always been one of those who had to work really hard to get decent grades. I am forever grateful to some of the amazing teachers that I have had who made learning one, and owing to whom I was able to get some extraordinary grades and get into one of the most prestigious universities of the country. Inspired by those same teachers, I am to be like one of them - who never gives up on her students and always believe in them!
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Five proton NMR spectra are given here, together with molecular formulas. In each case, propose a structure that is consistent with the spectrum. (a) C4Hg02 Offset: 24 ppm 0 4 10 (ppm) 50Hz (b)...
-
Propose a structure that is consistent with each set of 1H NMR data. IR data is provided for some compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) C4H1OO 6 (ppm) Splitting 1.28 1.35...
-
Identify the following five compounds of H, N, and O. For each compound, write a Lewis structure that is consistent with the information given. a. All the compounds are electrolytes, although not all...
-
According to the Statute of Frauds, in order to be legally enforceable, a contract must be in writing, name the contracting parties, identify the subject matter of the contract, and Be for a legal...
-
What are four recommendations for designing a successful MSS?
-
When agreeing to a SPAC merger, what uncertainty does the target firm face that affects the effective terms of the deal?
-
Determine the critical buckling load for the column. The column material can be assumed rigid. P 22. 2 k www A
-
Delacroix Co. had 800 units of inventory at the beginning of March 2010. Other information about that beginning Work in Process Inventory is as follows: Direct labor costs were extremely high during...
-
Watch the video on the investigation of the homicide to complete the following sections on the case: 1. Write the background information on your case. Explain what happened? Make sure to identify the...
-
Jill Donahue is the supervisor of the telephone operators who handle emergency calls from citizens and dispatch police, firefighters, and ambulances. One of her objectives for the coming year is to...
-
Manny owns a local fast-food franchise. Angel runs it for him. So in this situation, Manny is the ____________ and Angel is the ________________. a. Free rider; entrepreneur. b. Agent; principal. c....
-
A principal is worried that her agent may not do what she wants. As a solution, she should consider: a. Commissions. b. Bonuses. c. Profit sharing. d. All of the above.
-
Simplify the expression. Rationalize the denominator when necessary. 60
-
Josie owns 800 shares of Innovative Marketing Strategies Inc. It currently has a share price of $87.00 per share. The board is considering a three for two stock split. How much is Josie's investment...
-
A lathe takes 12 minutes to changeover to a different part. It can turn 1200 parts/hour. Available capacity is 38 hours/week. Demand requires 34 hours/week to process all the different can produce....
-
You have received three shipments of iron ore of the following weights: 2852, 1578, and 1877 lb. There is an uncertain gives 36.28 0.04%, 22.68 0.03%, and 49.23 0.06%, respectively. You are to pay...
-
Selected T-accounts of Moore Company are given below for the just completed year: Raw Materials Manufacturing Overhead Debit Credit Debit Credit Balance 1/1 Debits 20,000 Credits 130,000 ? Debits...
-
Suppose the demand for fidget spinners (remember those?) is represented by the equation: D = 60-p, where p is price, and that supply is represented by the equation S-15+3p-w, where w is the wage rate...
-
The current system of rating passers in the National Football League is based on four performance components: completions, touchdowns, yards gained, and interceptions, as percentages of the number of...
-
Catherine (aged 42) and Johnson (aged 45) have been married for 12 years. Johnson is a project manager of an event company at a monthly salary of $55,000 with an additional one-month salary of...
-
Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing the product with acid. For example, (a) How would you prepare...
-
When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain. -H C HCH2H2CH C
-
How might you carry out the following selective transformations? One of the two schemes requires a protection step. (Recall from Section 19.5 that aldehydes are more reactive than ketones toward...
-
During Year 1, Chung Corporation earned $6,900 of cash revenue and accrued $3,800 of salaries expense. Required: (Hint. Record the events in general ledger accounts under an accounting equation...
-
Prepare Cash Budget from Budgeted Transactions Prepare a cash budget for the month ended May 31, 2019. Campton Company anticipates a cash balance of $89,000 on May 1, 2019. The following budgeted...
-
The monthly salaries for December and the year-to-date earnings of the employees of Bush Consulting Company as of November 30 follow. Determine the amount of Medicare tax to be withheld from each...

Study smarter with the SolutionInn App


