Rank the following anions in terms of increasing basicity:
Question:
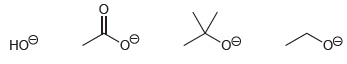
Transcribed Image Text:
ное
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Increa...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank the following anions in order of decreasing basicity: CH O CH,o CH
-
Draw all constitutional isomers with molecular formula C 2 H 6 S, and rank them in terms of increasing acidity.
-
For each of the following compounds, rank the highlighted bonds in terms of increasing wave number: a. b. TH. 0= H.
-
Mr. Paulo Rodriguez, a fund manager of The Blackstone Group Inc. holds a portfolio with a current value of RM7.83 million. The cash index currently stands at 1,075 points. He fears that the market...
-
Show that there are inputs that force every percolateDown in heapsort to go all the way to a leaf.
-
Austen Company manufactures and sells a single product. The companys sales and expenses for last year follow: Requirements 1. Fill in the missing numbers in the table. Use the table to answer the...
-
In the accompanying table, \(x\) is the tensile force applied to a steel specimen in thousands of pounds, and \(y\) is the resulting elongation in thousandths of an inch: (a) Graph the data to verify...
-
Rogers made a contract with Salisbury Brick Corp. that allowed it to remove earth and sand from land he owned. The contract ran for four years with provision to renew it for additional four-year...
-
Evaluate the challenges and strategies associated with implementing quality control in complex, global supply chains, considering the impact of multi-tier suppliers, geographical dispersion, and...
-
As T.C. Resort Properties seeks to improve its customer service, the company faces new competition from SunLow Resorts. SunLow has recently opened resort hotels on the islands where T.C. Resort...
-
Which of the following is not one of the four major economic flows linking the U.S. economy with that of other nations? a. Trade flows. b. Resource flows. c. Financial flows. d. Foreign aid flows.
-
Compare a hypothetical DVC with a hypothetical IAC. In the DVC, average per capita income is $500 per year. In the IAC, average per capita income is $40,000 per year. If both countries have a savings...
-
In Yukawas original theory (1934), which remains a useful approximation in nuclear physics, the strong force between protons and neutrons is mediated by the exchange of -mesons. The potential energy...
-
Based on the DuPont analysis, Abays belief regarding ROE is most likely based on: A. leverage. B. profit margins. C. asset turnover. Quentin Abay, CFA, is an analyst for a private equity firm...
-
Based on Ohalins estimates, the amount of the joint ventures 31 December 2010 total assets (in $ millions) that will be included on Supreme Healthcares consolidated financial statements will be...
-
To account for its foreign operations, Romulus has most likely designated the euro as the functional currency for: A. Julius only. B. Augustus only. C. both Julius and Augustus. Romulus Corp. is a...
-
If the US dollar were chosen as the functional currency for Acceletron in 2007, Redline could reduce its balance sheet exposure to exchange rates by: A. selling SGD30 million of fixed assets for...
-
Will the change in accounting rules impact the result of the initial screening process for Colorful Concepts? A. It passes the screens now, but will not pass if the accounting rules change. B. It...
-
Sketch a graph of a function f that is continuous on (-, ) and has the following properties. f'(x) > 0, f"(x) > 0
-
What is EBIT/eps analysis? What information does it provide managers?
-
Tell the number of hydrogens bonded to each carbon atom in the following substances and give the molecular formula ofeach: OH H (a) (b) CO2CH3 Ephedrine Cocaine
-
Identify the most electronegative element in each of the following molecules: (a) CH2FC1 (b) FCH2CH2CH2Br (c) HOCH2CH2NH2 (d) CH3OCH2Li
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
Explain possible advantages and disadvantages of PSL's current international trading strategy (subsidiary in China, export & import and online presence).
-
In November 2020 you entered into four May 2021 long futures contracts for crude oil (1,000 barrels per contract) for $65.00 per barrel. If the futures price was $70.00 per barrel on December 31,...
-
4) Champagne, Inc., had revenues of $13 million, cash operating expenses of $9 million, and depreciation and amortization of $2 million during 2008. The firm purchased $650,000 of equipment during...

Study smarter with the SolutionInn App


