For each of the following compounds, rank the highlighted bonds in terms of increasing wave number: a.
Question:
a.
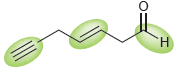
b.
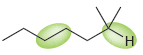
Transcribed Image Text:
TH. 0= H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a b In...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds and ions, 1. Draw a Lewis structure. 2. Show the kinds of orbitals that overlap to form each bond. 3. Give approximate bond angles around each atom except...
-
For each of the following compounds, draw the form in which it will predominate at pH = 3, pH = 6, pH = 10, and pH = 14: a. b. c. CH3COOH pKa = 4.8 CH3CH2NH3 pKa 11.0 CF CH2OH pKa 12.4
-
Suppose you buy Bond A at time=0. It is a 2-year, risk-free bond with coupon rate of 7% that compounds annually, and it has a face value of $1,000. (3 points) a) At time = 0, suppose you buy Bond A...
-
The inner loop of the cubic maximum subsequence sum algorithm performs N(N+1)(N+2)/6 iterations of the innermost code. The quadratic version performs N(N + 1)/2 iterations. The linear version...
-
Use the longevity times for presidents and construct a 95% confidence interval estimate of the population mean. Presidents 10 29 26 28 15 23 17 25 0 20 4 1 24 16 12 4 10 17 16 0 7 24 12 4 18 21 1 2 9...
-
Two streams, each containing \(0.5 \mathrm{lb} / \mathrm{hr}\) steam at \(550 \mathrm{psia}\), are mixed as shown in Figure 10.29: (a) Compute the heat loss to an environmental reservoir at...
-
An investor purchased the following 5 bonds. Each bond had a par value of $1,000 and an 8% yield to maturity on the purchase day. Immediately after the investor purchased them, interest rates fell...
-
Crane Company has several processing departments. Costs to be accounted for in the Assembly Department for November 2022 totaled $2,268,894 as follows. Work in process, November 1 Materials $78,800...
-
The diagram shows a semi-circle with diameter EF of length 12 cm. Angle GEF = radians and the shaded region has an area of A cm 2 . a. Show that A = 36 + 18 sin 2. b. Given that is increasing at a...
-
If a woman does not get an interview for a job requiring heavy lifting because the manager has noted that the average women can lift less than the average man, this is a. A legal example of...
-
The argument that casinos have little economic impact on a community is based on the notion of a. Supply. b. Demand. c. Opportunity cost. d. Local substitution.
-
What is spend analysis, and what questions can it help answer?
-
For the following examples, state what the correct value is and what accounting concept justifies your answer: a. NYU Health bought a magnetic resonance imaging (MRI) machine worth $1,500,000, but...
-
Why might too much liquidity be a problem for an organization? Why might too little be a problem?
-
What are the steps in managing the revenue cycle?
-
What are the three primary reasons an organization holds cash or cash equivalents?
-
How much will $6,000 invested at 5% simple interest be worth in 3 years? What will it be worth if the interest rate is 7%?
-
Develop a group response to the following question: Can your team identify other solutions to this dilemma?
-
Determine the values of the given trigonometric functions directly on a calculator. The angles are approximate. tan 0.8035
-
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain why none of the other regioisomer isproduced. Br + HBr
-
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a mechanism for thereaction. H. + CHCI3 AICI3
-
At what position, and on what ring, would you expect the following sub stances to undergo electrophilicsubstitution? (a) (b) CH3 Br (c) (d) .CI CH3 Z O=U
-
The Clifford Corporation has announced a rights offer to raise $36 million. The stock currently sells for $28 per share and there are 18 million shares outstanding. If the subscription price is set...
-
Superman Enterprises has just completed an initial public offering. The firm sold 4,700,000 new shares at an offer price of $17.00 per share. The underwritering spread was $0.78 a share. The firm...
-
ABV is a non-dividend paying stock whose current price is $50. Its volatility is 12%. Over each of the next two 6-month periods the stock price is expected to go up by 9% or down by 8%. The risk-free...

Study smarter with the SolutionInn App


