(R)-Limonene is found in many citrus fruits, including oranges and lemons: Draw the structures and identify the...
Question:
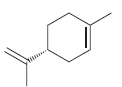
Draw the structures and identify the relationship of the two products obtained when (R)-limonene is treated with excess hydrogen in the presence of a catalyst.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
exces...View the full answer

Answered By

PU Student
cost accounting
financial accounting
auditing
internal control
business analyst
tax
i have 3 years experience in field of management & auditing in different multinational firms. i also have 16 months experience as an accountant in different international firms. secondary school certification.
higher secondary school certification.
bachelors in mathematics.
cost & management accountant
4.80+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(R)-Limonene is found in many citrus fruits, including oranges and lemons: For each of the following compounds identify whether it is (R)-limonene or its enantiomer, (S)-limonene: a. b. c. d.
-
Tartaric acid is found in many fruits, including grapes, and is partially responsible for the dry texture of certain wines. Calculate the pH and the tartarate ion (C4H4O62-) concentration for a 0.250...
-
Citric acid (C6H8O7) is the acid found in citrus fruits. It can lose a maximum of three H+ ions in the presence of a base. Write the chemical equations for citric acid acting stepwise as a...
-
Find steady state current i of a RL circuit with L=2 H and R=892 which can be modeled by the following differential equation: 2di/dt+8i=v(t) where v(t) = 16 cos(10t -45) with phasor technique. Also...
-
a. To what extent can we really see the world as it really is? b. To what extent is truth a function of power? c. To what extent to texts have stable meanings? d. What are the best Christian...
-
How many user names are possible in Problem 41 if the last character must be a digit?
-
Consider the simulation flowsheets in Figure 7.36, which were prepared for ASPEN PLUS. The feed stream, S1, is specified, as are the parameters for each process unit. Complete the simulation...
-
Kroger, Safeway Inc., and Winn-Dixie Stores Inc. are three grocery chains in the United States. Inventory management is an important aspect of the grocery retail business. Recent balance sheets for...
-
Paypal was rumored to be in talks to acquire Pinterest for a 25% premium. Assume that the two companies reached agreement on that basis. Using the stock prices given below (assume that the premium...
-
ABC Framing has been hired to frame a light commercial building. The project began on July 2 and was completed on August 9. The following is a list of accounting transactions associated with the...
-
When engaging in monetary policy, the impact of expansionary policy on an aggregate demand aggregate supply model is to a. Increase aggregate demand. b. Increase aggregate supply. c. Decrease...
-
The elasticity of demand is related to the slope of the demand curve A. And only the slope of the demand curve. B. But also the (price, quantity) position on the demand curve. C. But also the slope...
-
Discuss the differences in how people react to change over time. What does the research based on this model suggest organizations do when implementing change programs?
-
Use the data from question 13 to produce a cumulative frequency graph and a cumulative relative frequency graph. Question 13 An advertising executive is interested in the age distribution of the...
-
What can fill in the blank so the play() method can be called from all classes in the com.mammal.eland package, but not the com.mammal.gopher package? package com.mammal; A. Leave it blank B. private...
-
L and M form the LM Partnership with equal cash contributions of $300,000. The partnership then borrows $1,400,000 and purchases several tracts of land at a total cost of $1,900,000. Immediately...
-
Construct a relative frequency histogram. Use the following information to answer question. Suppose a class of high school seniors had the following distribution of SAT scores in English. SAT score...
-
The light bulbs produced by Edison Lighting Corporation last an average of 300 h. The life of the light bulbs is believed to follow a normal distribution with a standard deviation of 10. A customer...
-
In terms of limits, what does it mean for f to grow faster than g as x?
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
Explain which compound has the higher melting point or boiling point: a) Melting point b) Boiling point or C COCH c) Boiling point or
-
What is the functional group present in these compounds? SCH3 SH C CH, - Ph b) ) H3 f) CH,CH,SOH d) CH,CH,SPh e) CH,SCH3
-
Identify the most acidic site in thesecompounds: NH2 CH.COCH,CH,CH3 c) a) b) CH3 e) CH;CH,CH,COH d) CH,CH,CCH,CH3
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...
-
The trial balance for a company listed the following account balances at December 31, Year 1, the end of its fiscal year: cash, $36,000; accounts receivable. $31,000; Inventory, $45,000; equipment...

Study smarter with the SolutionInn App


