Starting with acetylene as your only source of carbon atoms, propose a plausible synthesis for 1, 4-dioxane:
Question:
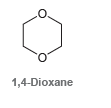
Transcribed Image Text:
1,4-Dioxane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
H Lindlars ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using acetylene as your only source of carbon atoms, design a synthesis of trans-5- decene:
-
Using acetylene as your only source of carbon atoms, design a synthesis of cis-3- decene:
-
Using acetylene as your only source of carbon atoms, design a synthesis of pen-tanal. (Pentanal has an odd number of carbon atoms, while acetylene has an even number of carbon atoms): H.
-
East Corp is a mature company and expects its sales and operating costs to be fixed (or unchanged) in perpetuity. It has sales of $524,000 and operating costs of $272,480. The firm has a tax rate of...
-
a. In how many ways can two sorted arrays of N elements be merged? b. Give a nontrivial lower bound on the number of comparisons required to merge two sorted lists of N elements, by taking the...
-
Happy Giant produces canned chicken à la king. The chicken à la king passes through three departments: Mixing, Retort (sterilization), and Packing. In the Mixing Department, chicken and...
-
With reference to the preceding exercise, find \(99 \%\) limits of prediction for the level of air pollution when the flow of vehicles is \(30 \%\). Also indicate to what extent the width of the...
-
Cray Company started year 2 with $60,000 in its cash and common stock accounts. During year 2 Cray paid $45,000 cash for employee compensation. Assume this is the only transaction that occurred in...
-
John wants to be able to withdraw $10,426 every year for 14 years from his retirement fund 1 year after he retires. How much he must accumulate in his retirement account if the interest rate is 4%...
-
Arnoff Enterprises manufactures the central processing unit (CPU) for a line of personal computers. The CPUs are manufactured in Seattle, Columbus, and New York and shipped to warehouses in...
-
Assume a DVC and an IAC presently have real per capita outputs of $500 and $5000, respectively. If both nations have a 3 percent increase in their real per capita outputs, by how much will the per...
-
True or False: Some economists argue that the single best thing that IACs could do for DVCs in terms of economic growth would be to eliminate trade barriers between IACs and DVCs.
-
Elliot Willensky and Beverly Moran formed a partnership to renovate and flip (resell) some property. According to their agreement, Moran would finance the purchase and renovation of the property, and...
-
When one hears the word entrepreneur, many faces come to mind. Elon Musk, Sara Blakely, Warren Buffet, Martha Stewart, Bill Gates, and Daymond John are just a few that might spring to mind. There are...
-
An organization chart is a diagram showing how employees and tasks are grouped and how the lines of communication and authority flow within an organization. These charts can look very different...
-
Over the past few years, employees have been expected to function as productive team members instead of working alone. People often believe that they can work effectively in teams, but many people...
-
Today, firms around the world need employees with special skills. In some countries, such employees are not always available, and firms then must search abroad for qualified applicants. One way they...
-
Why should small businesses engage in exporting?
-
Evaluate the following limits. Check your results by graphing. lim x/x -
-
Using a graphing utility, graph y = cot -1 x.
-
Imidazole forms part of the structure of the amino acid histidine and can acts as both an acid and a base (a) Look at the electrostatic potential map of Imidazole, and identify the most acidic...
-
Of the two vitamins A and C, and is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which iswhich? CH CH- CH CH2OH H3C CH3 H- CH Vitamin A (retinol) Vitamin C...
-
Fill in the multiple bonds in the following model of naphthalene, C10H8 (gray = C, ivory =H). How many resonance structures does naphthalenehave?
-
1). The Electronic Corporation manufactures two electrical products: air conditioners and large fans. The assembly process for each is similar in that both require a certain amount of wiring and...
-
After reviewing oceanview marine company's case materials, and reviewing the unaudited account balances, what is your preliminary view on the client's ability to continue as a going concern? What is...
-
For each chemical reaction listed in the table below, decide whether the highlighted atom is being oxidized or reduced. reaction highlighted atom is being... neither oxidized reduced oxidized nor...

Study smarter with the SolutionInn App


