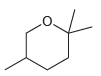
The following cyclic ether can be prepared via an intramolecular Williamson ether synthesis. Show what reagents you
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Ho...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. HO. HO.
-
Which of the following is not necessary to do before you can run a Java program? a. Coding b. Compiling c. Debugging d. Saving
-
Salmon Inc. has debt with both a face and a market value of $3,000. This debt has a coupon rate of 7% and pays interest annually. The expected earnings before interest and taxes is $1,200, the tax...
-
A first-time shareholder has approached you requesting some advice. The shareholder has received the companys annual report and noticed the following statement in the summary of significant...
-
If the maximum allowable bending stress for the \(\mathrm{W} 10 \times 15\) structural A992 steel beam is \(\sigma_{\text {allow }}=20 \mathrm{ksi}\), determine the maximum height \(h\) at which a...
-
Kendal County engaged in the following transactions. For each, prepare an appropriate journal entry and indicate the type of fund in which it would most likely be recorded: 1. It levied and collected...
-
What are the similarities or differences between the revivals of the 18th to 19th centuries to the ones we see today ?
-
Describe each of the following situations in the language of options. State in each case whether the situation involves a call or a put and the option's exercise price. a. A company has drilling...
-
The heat capacity of α -quartz is given by The coefficient of thermal expansion is given by β = 0.3530 à 10 4 K 1 and V m = 22.6 cm 3 mol+. Calculate ÎS m for...
-
Does the enthalpy of formation of H 2 O(l) change if the absolute enthalpies of H 2 (g) and O 2 (g) are set equal to 100. kJ mol 1 rather than to zero? Answer the same question for CO 2 (g). Will H o...
-
According to a recent Current Population Survey, the mean personal income for American adults was $38,337. A histogram for incomes is skewed to the right. Which of the following is a possible value...
-
A major retailer promises 2 day shipping for certain online orders. A consumer advocacy group believes that the shipping times are longer at a significance level of 0.05, and checks a randomly chosen...
-
The sales tax rate in Temecula, California is 8.75%. A sporting goods store collected $73,925.18 in sales taxes in the month of July. How much were their total sales (not including the sales tax...
-
Explain why you think there are so many different types of gender theories .
-
What marketing strategy and plan would you recommend for the new fitness tracking device startup company to generate awareness and drive sales with limited resources?
-
What are some strategies for building quality into projects? What are some types of documentation that must be maintained for projects? What are the characteristics of good documents?
-
Write an equation for each conic section. Parabola with vertex at the origin; through the point (10, -5); opens down
-
Evenflow Power Co. is considering a new project that is a little riskier than the current operations of the company. Thus, management has decided to add an additional 1.5% to the company's overall...
-
Amine bases in nucleic acids can react with alkylating agents in typical SN2 reactions. Look at the following electrostatic potential maps, and tell which is the better nucleophile, guanine or...
-
Human brain natriuretic peptide (BNP) is a small peptide of 32 amino acids used in the treatment of congestive heat failure. How many nitrogen bases are present in the DNA that codes for BNP?
-
Human and horse insulin both have two polypeptide chains, with one chain containing 21 amino acids and the other containing 30 amino acids. They differ in primary structure at two places. At position...
-
Calculate Ending Inventory using Weighted average (WA) method Syd Gee Ltd Opening Trial Balance 1 June 2021 BANK STATEMENT As at 30 June 2021 DATE PARTICULARS DEBIT CREDIT BALANCE Account Debit...
-
A skier glides down a frictionless hill of 100 meters high, then ascends another hill, with a height of 90 meters, as shown in the figure below. a) What is the speed of the skier when they reach the...
-
The atoms in a nickel crystal vibrate as harmonic oscillators with an angular frequency of 5.5x 1013 rad/s. The mass of a nickel atom is 9.75 x 10-26 kg. What is the difference in energy between...

Study smarter with the SolutionInn App


