The heat capacity of α -quartz is given by The coefficient of thermal expansion is given by
Question:
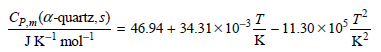
The coefficient of thermal expansion is given by β = 0.3530 × 10ˆ’4 Kˆ’1 and Vm = 22.6 cm3 mol+. Calculate ΔSm for the transformation α -quartz (15.0°C,1atm) †’α -quartz (420.°C, 925 atm).
Transcribed Image Text:
Cp m (a-quartz, s) Jк'mol- 46.94 + 34.31x10-3 - 11.30 x10°, к? к
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
ASm I Cpm VBPs P dT T 69315 28815 46943431101 113x105 10 1 ...View the full answer

Answered By

Saikumar Ramagiri
Financial accounting:- Journal and ledgers, preparation of trail balance and adjusted trail balance Preparation of income statement, retained earning statement and balance sheet Banks reconciliation statements Financial statement analysis Cash flow statement analysis (both direct and indirect methods) All methods of Depreciations Management Accounting:- Ratios Budgeting control Cash budget and production budget Working capital management Receivable management Costing:- Standard and variance costing Marginal costing and decision making Cost-volume-profit analysis Inventory management (LIFO, FIFO) Preparation and estimation of cost sheet Portfolio management:- Calculation of portfolio standard deviation or risk Calculation of portfolio expected returns CAPM, Beta Financial management:- Time value of money Capital budgeting Cost of capital Leverage analysis and capital structure policies Dividend policy Bond value calculations like YTM, current yield etc International finance:- Derivatives Futures and options Swaps and forwards Business problems Finance problems Education (mention all your degrees, year awarded, Institute/University, field(s) of major): Education Qualification Board/Institution/ University Month/Year of Passing % Secured OPTIONALS/ Major ICWAI(inter) ICWAI inter Pursuing Pursuing - M.com(Finance) Osmania University June 2007 65 Finance & Taxation M B A (Finance) Osmania University Dec 2004 66 Finance & Marketing. B.Com Osmania University June 2002 72 Income Tax, Cost & Mgt, Accountancy, Auditing. Intermediate (XII) Board of Intermediate May 1999 58 Mathematics, Accountancy, Economics. S S C (X) S S C Board. May 1997 74 Mathematics, Social Studies, Science. Tutoring experience: • 10 year experience in online trouble shooting problems related to finance/accountancy. • Since 6 Years working with solution inn as a tutor, I have solved thousands of questions, quick and accuracy Skills (optional): Technical Exposure: MS Office, SQL, Tally, Wings, Focus, Programming with C Financial : Portfolio/Financial Management, Ratio Analysis, Capital Budgeting Stock Valuation & Dividend Policy, Bond Valuations Individual Skills : Proactive Nature, Self Motivative, Clear thought process, Quick problem solving skills, flexible to complex situations. Achievements : 1. I have received an Award certificate from Local Area MLA for the cause of getting 100% marks in Accountancy during my Graduation. 2. I have received a GOLD MEDAL/Scholarship from Home Minister in my MBA for being the “Top Rank student “ of management institute. 3. I received numerous complements and extra pay from various students for trouble shooting their online problems. Other interests/Hobbies (optional): ? Web Surfing ? Sports ? Watching Comics, News channels ? Miniature Collection ? Exploring hidden facts ? Solving riddles and puzzles
4.80+
391+ Reviews
552+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The heat capacity of anhydrous potassium hexacyanoferrate (II) varies with temperature as follows: TIK Cp,m/J K-I mol-I) TIK Cp,m/JK-I mol-I) 10 2.09 100 179.6 20 14.43 110 192.8 30 36.44 150 237.6...
-
Two formulas for the heat capacity of CO are given here: Cp [cal/(mol C)] = 6.890 + 0.001436T (C) Cp (Btu/(lb-moleF)] = 6.864 + 0.0007978T (F) Starting with the first formula, derive the second....
-
A small pharmaceutical firm plans to manufacture a new drug and has hired you as a consultant to design a condenser to remove the drug from a gasvapor mixture. The mixture, which contains 20 mole% of...
-
Wical Rental Management Services manages four apartment buildings, each with a different owner. Wicals CEO has observed that the apartment buildings with more expensive rental rates tend to require...
-
Spice Inc.'s unit selling price is $60, the unit variable costs are $35, fixed costs are $125,000 and current sales are 10,000 units. How much will operating income change is sales increase by 8,000...
-
The general ledger trial balance of David Ltd at 30 June 2022 includes the following asset and liability accounts. The lease liability (k) includes an amount of \($26\) 000 for lease payments due...
-
A 40-lb weight is dropped from rest at a height of \(h=2 \mathrm{ft}\) onto the center of the W10 \(\times 15\) structural A992 steel beam. Determine the vertical displacement of its end \(B\) due to...
-
Presented below are a number of independent situations. Instructions For each individual situation, determine the amount that should be reported as cash. If the item(s) is not reported as cash,...
-
what does a center proposal looks like. Planned center, how will you assess the center, what outcomes will children achieve?, How will you assess the outcomes? timeline for making decisions, and...
-
The tensile strength of paper is modeled by a normal distribution with a mean of 35 pounds per square inch and a standard deviation of 2 pounds per square inch. (a) What is the probability that the...
-
Calculate the average CH bond enthalpy in methane using the data tables. Calculate the percent error in equating the average CH bond energy in Table 4.3 with the bond enthalpy. Table 4.3 Selected...
-
The following cyclic ether can be prepared via an intramolecular Williamson ether synthesis. Show what reagents you would use to make this ether.
-
Fill in each statement with the appropriate capital investment analysis method: Payback, ARR, NPV, or IRR. Some statements may have more than one answer. a. ______ is ( are) more appropriate for...
-
The latitude of a city is 25 N, and the latitude of the north end of a canal is 6 N. Both are at a longitude of 71 W. What is the distance between the city and the canal? Explain how the angle used...
-
Why is it important to identify and reflect on your own social and cultural perspectives and biases?
-
13. Consider the following code: public class Point { private int x; } private int y%;B public Point() { this.x 0; this.y = 0%; } //Point public class PointRunner { } public static void main(String...
-
We have said that todays professional sales representative is a marketing consultant and a manager of a market- his or her territory. Explain how a sales rep. can be a marketing consultant and...
-
Who are the major parties involved in this issue? Which market forces have an impact on this issue? How is demand illustrated in this issue? How is supply illustrated in this issue? How has the...
-
Write an equation for each conic section. Ellipse with center (3, -2); a = 5; c = 3; major axis vertical
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
In which of the following systems is the energy level separation the largest? (a) A 14Nnucleus in (for protons) a 600 MHz NMR spectrometer, (b) An electron in a radical in a field of 0.300 T
-
Calculate the magnetic field needed to satisfy the resonance condition for unshielded protons in a 150.0 MHz radiofrequency field.
-
Use Table 15.2 to predict the magnetic fields at which (a) 14N, (b) 19F, and (c) 31p comes into resonance at (i) 300 MHz, (ii) 750 MHz.
-
On December 31, Jarden Company's Allowance for Doubtful Accounts has an unadjusted credit balance of $15,000. Jarden prepares a schedule of its December 31 accounts receivable by age. Age of Accounts...
-
Recently, a famous movie star won a defamation trial against his ex-wife, also a famous movie star. The ex-husband claimed that his ex-wife had defamed him in a Washington Post column, where she...
-
On 1 August 2022, Jack, with the written approval of Mega Phones Board, then approaches Jill from Techworld to supply 500 Apple iPhones Mark XYZ, which have recently been released, for a total price...

Study smarter with the SolutionInn App


