When the following optically active alcohol is treated with HBr, a racemic mixture of alkyl bromides is
Question:
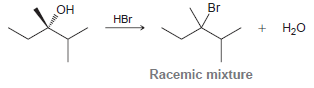
Draw the mechanism of the reaction, and explain the stereochemical outcome.
Transcribed Image Text:
Br Он HBr + H20 Racemic mixture Мн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
The chirality center is lost when the leaving g...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When 2-phenyl-2-butanol is allowed to stand in ethanol containing a few drops of sulfuric acid, the following ether is formed: Suggest a reasonable mechanism for this reaction based on the...
-
Compound A is an optically active alcohol. Treatment with chromic acid converts A into a ketone, B. In a separate reaction, A is treated with PBr3, converting A into compound C. Compound C is...
-
When a primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out...
-
You're pretty sure that your candidate for class president has about 55% of the votes in the entire school. But you're worried that only 100 students will show up to vote. How often will the underdog...
-
What is WBGT?
-
Consider the emergence of social class and the emergence of the way we perceive ourself. Discuss if it is an example of two interacting emergent phenomena, as seems to be implied in [277].
-
The phase change process at varied equilibrium pressure and temperature is concerned with (a) Maxwell's equation (b) Gibbs-Helmholtz equation (c) Clapeyron equation (d) Gibbs-Duhem equation.
-
Pataky Co.'s sales manager estimates that 2,000,000 units of product RI#698 will be sold in 2011. The product's selling price is expected to decline as the result of technology changes during the...
-
Former US President Trump is starting to draft an economic plan for his 2024 presidential campaign. He wants to keep the tax cuts implemented by the Trump administration in 2017, the Tax Cuts and...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
What kind of food did the extinct equid likely eat?
-
When ethylbenzene is treated with NBS and irradiated with UV light, two stereoisomeric compounds are obtained in equal amounts. Draw the products and explain why they are obtained in equal amounts....
-
The small piston of a hydraulic lift has a cross-sectional area of 3.00 cm2, and its large piston has a cross-sectional area of 200 cm2 (Figure 14.4). What force must be applied to the small piston...
-
A 9.98% coupon, 14.0 -year annual bond has a yield to maturity of 7.57%. Assuming the par value is 1,000 and the YTM does not change over the next year, Compute the following: A. Price of the bond...
-
Whirly Corporation's contribution format income statement for the most recent month is shown below: Sales (7,300 units) Variable expenses Contribution margin Fixed expenses Total $ 219,000 146,000...
-
Most home insurance policies cover jewellery and silverware for a limited amount unless items are covered with additional insurance via an umbrella plan. If $17,500 worth of jewellery and $3,750...
-
Describe an example of how formal or informal mediation might be used to facilitate Ottos rights.
-
What does this mean exactly: No. In order to claim a Military Benefits Subtraction (also known as the Military Retirement Subtraction), the eligible military benefits subtracted must be included as...
-
Points at which the graphs of r = f() and r = g() intersect must be determined carefully. Solving f() = g() identifies somebut perhaps not allintersection points. The reason is that the curves may...
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
Predict the geometry at the carbon of these compounds: a) H-C=N: b) 0: H-C-0-H c) H T CI-C-H T CI
-
Predict the geometry of the following compounds at the indicated atoms: a) H H-C-0-H H At the carbon At the oxygen b) H. H C-N-H At the carbon At the nitrogen c) H. C=O-H H At the carbon At the oxygen
-
Predict the direction of the dipole moments of these compounds: a) H H b) F H-C-H H H H-C-C=N H
-
A manufacturer reports finished goods inventory of $820 on June 1 and $1,130 on June 30. Cost of goods manufactured for June is $5,460. What is cost of goods sold for June? Cost of Goods Sold Cost of...
-
Stacey's Piano Rebuilding Company has been operating for one year. At the start of the second year, its income statement accounts had zero balances and its balance sheet account balances were as...
-
MyBnB started a home rental company on January 1. As of November 30, MyBnB reported the following balances. The company does not yet have a balance in Retained Earnings because this is its first year...

Study smarter with the SolutionInn App


