The data in the following table have been obtained for the potential of the cell Pt(s) |H
Question:
The data in the following table have been obtained for the potential of the cell Pt(s) |H2(g, f = 1atm) HCl(aq, m)|AgCl(s) Ag(s) as a function of m at 25°C.
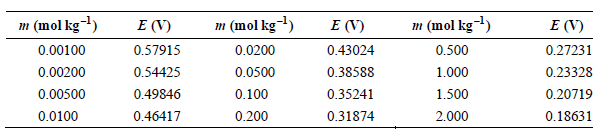
a. Determine E° using a graphical method.
b. Calculate γ ± for HCl at m = 0.00100, 0.0100, and 0.100 mol kg–1.
Transcribed Image Text:
m (mol kg) m (mol kg) m (mol kg-l) E (V) 0.57915 0.54425 0.49846 E (V) E (V) 0.27231 0.23328 0.20719 0.500 0.00100 0.0200 0.43024 0.38588 0.35241 0.00200 0.0500 1.000 0.00500 0.0100 1.500 2.000 0.100 0.200 0.46417 0.31874 0.18631
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
Cell reaction 2AgCls H 2 g2Ags 2H aq 2Cl aq In the low ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following data have been obtained for oxygen gas at 273.15 K. Calculate the best value of the gas constant R from them and the best value of the molar mass of02 0.750000 0.250000 Platm 0.500000...
-
The following data have been obtained for the liquid-vapour equilibrium compositions of mixtures of nitrogen and oxygen at 100 kPa. T/K 77.3 78 80 82 84 86 88 90.2 X (O2) 0 10 34 54 70 82 92 100...
-
Your task in this problem will be to use a spreadsheet to generate a Txy diagram for a two-component system, using Raoult?s law to express the vapor?liquid equilibrium distribution of each species....
-
A problem in Statistics is given to three students A, B, and C whose chances of solving it are 1/4 and 1/5 respectively. Find the probability that the problem will be solved if they all try...
-
Discuss the key ethical issues of taste, stereotyping, use of sex appeal, and targeting children. How do advertising professionals deal with these issues?
-
Table B. 2 presents data collected during a solar energy project at Georgia Tech. a. Fit a simple linear regression model relating total heat flux $y$ (kilowatts) to the radial deflection of the...
-
Fill in the Blank. An active isolator consists of a sensor, a signal processor, and a(n) ____________ .
-
The stockholders' equity section of Lachlin Corporation's balance sheet at December 31 is presented here. Instructions From a review of the stockholders' equity section, answer the following...
-
Analyse the financial statements of Fisher &Paykel Healthcare Limited (FPH) and KMD Brands (KMD) for the latest financial years. You may use the following links to download the companies' annual...
-
An experiment was conducted to test the efficacy of chloromycetin in checking typhoid. In a certain hospital chloromycetin was given to 285 out of the 392 patients suffering from typhoid. The number...
-
Show that the estimates Î²Ì 1 = 1 . 572 and Î²Ì 2 = 1 . 357 used in the first experiment of Table 3.1 are in fact the OLS estimators. Table 3.1 Y2i (6) Y; X (2) i...
-
What is the mean of X? One interesting phenomenon of bacteriuria is that there is a turnover; that is, if bacteriuria is measured on the same woman at two different points in time, the results are...
-
"In-house inventors are tough to deal with. Right now we have this Ph.D. in physics, a really great person, bright as they come, and terribly creative. Has had no less than 11 ideas go to market...
-
Ingrid has configured one of the network segments to use a DHCP server to dynamically assign IP addresses. She knows that DHCP can also tell the clients the address of the server that they should use...
-
Your company is excited with the information discussed regarding the cloud technologies that are available to Get-Sole Shoes. Now the management team wants to examine what its cloud infrastructure...
-
A corporate bond with a face value of $1,000 and a coupon rate of 4.6% pays interest semiannually and has a maturity date of May 6, 2026. The trade settles on January 22, 2022. The yield to maturity...
-
Bodin Company manufactures finger splints for kids who get tendonitis from playing video games. The firm had the following inventories at the beginning and end of the month of January. January 1...
-
How do cognitive biases, such as confirmation bias and anchoring, impact decision-making during conflict resolution, and what strategies can be employed to mitigate their effects and promote more...
-
Evaluate the following integrals analytically. sinh -1 x dx
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Discuss the physical origins of the surface Gibbs energy.
-
The rate of sedimentation of a recently isolated protein was monitored at 20C and with a rotor speed of 50 000 r.p.m. The boundary receded as follows: Calculate the sedimentation constant and the...
-
Find the drift speed of a particle of radius 20 m and density 1750 kg m 3 which is settling from suspension in water (density 1000 kg m 3 ) under the influence of gravity alone. The viscosity of...
-
Matrix B is given as follows, -18 20 -61 B-12 14 -3 30 -30 13. a) Use the Gerschgorin's Circle Theorem to determine a region containing all the eigenvalues of B. b) Find the dominant eigenvalue (21)...
-
5.63. Acetone can be removed from acetone-air mixtures using simple counter- current cascades, by adsorption onto charcoal (Foust et al. 1980). We wish to find the required number of equilibrium...
-
Use characteristic polynomial to find the eigen value and eigen vector for A S

Study smarter with the SolutionInn App


