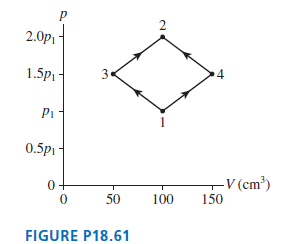
FIGURE P18.61 shows two different processes by which 1.0 g of nitrogen gas moves from state 1
Question:
FIGURE P18.61 shows two different processes by which 1.0 g of nitrogen gas moves from state 1 to state 2. The temperature of state 1 is 25?C. What are
(a) Pressure p1
(b) Temperatures (in ?C) T2, T3, and T4?
Transcribed Image Text:
2.0p 1.5p 3. Pi 0.5p1 - V (cm³) 50 100 150 FIGURE P18.61 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Model Assume the nitrogen gas is an ideal gas Solve a ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
FIGURE P18.62 shows two different processes by which 80 mol of gas move from state 1 to state 2. The dashed line is an isotherm. a. What is the temperature of the isothermal process? b. What maximum...
-
Figure Q19.7 shows two different processes taking an ideal gas from state i to state f. Is the work done on the gas in process A greater than, less than, or equal to the work done in process B?...
-
Figure Q19.8 shows two different processes taking an ideal gas from state i to state f. a. Is the temperature change ÎT during process A larger than, smaller than, or equal to the change during...
-
DS Unlimited has the following transactions during August. August 6 Purchases 88 handheld game devices on account from GameGirl, Inc., for $290 each, terms 1/10, n/60. August 7 Pays $490 to Sure...
-
Blonde Corporation has a calendar year retirement plan covering its participants. William, age 21, is hired on January 31, 2015, and he meets the service requirement over the next 12 months...
-
Again consider the domain model class diagram shown in Figure 4-16. Add the following to the diagram and list any assumptions you had to make: A faculty member usually teaches many course sections,...
-
The suitable pump for flood control and irrigation applications is (a) reciprocating (b) centrifugal (c) axial flow (d) mixed flow
-
A state requires large merchants (i.e., those with sales over a specied dollar amount) to report and remit their sales taxes within 15 days of the end of each month. It requires small merchants to...
-
Explain what is an organizational code of ethics (Ch 3 topic)? Why should organizations have a code of ethics? Explain the "three nudge systems" discussed in the media clip. Explain why or why not,...
-
A composite wall separates combustion gases at 2600C from a liquid coolant at 100C, with gas- and liquid-side convection coefficients of 50 and 1000 W/m 2 K. The wall is composed of a 10-mm-thick...
-
Selected data for The Hershey Company for Year 1 through Year 3 appear in Exhibit 4.29. REQUIRED a. Compute ROA and its decomposition for Year 2 and Year 3. Assume a tax rate of 35%.b. Compute ROCE...
-
Hasbro is a leading firm in the toy, game, and amusement industry. Its promoted brands group includes products from Playskool, Tonka, Milton Bradley, Parker Brothers, Tiger, and Wizards of the Coast....
-
A portfolio manager considers the following annual coupon bonds: An increase in expected inflation causes the government yield curve to steepen, with a 20-point rise in the 10-year government bond...
-
Law on Partnership ( Arts 1 7 6 7 up to 1 8 6 7 of the New Civil Code ) 1 . DEFINITION OF PARTNERSHIP under art 1 7 6 7 of the new Civil Code 2 . CHARACTERISTIC ELEMENTS OF A CONTRACT OF PARTNERSHIP...
-
What are the duties that must be fulfilled at all times when providing Financial Advice to a client?
-
Are they the pressures and series of events that led WorldCom executives and managers to "cook the books"? Explain in detail. Explain in detail the setting and all the events that led to the...
-
Intermittent flow is experienced from a cylindrical silo with a conical hopper that is several years old. The design criteria have been checked by re-measuring the powder properties and wall...
-
The CEO of a company would like to offer free parking to all employees in a prime downtown space. What impact will this have on their pay?
-
Based on the evaluation you developed for Question 3, provide responses to the ethical decision-making metric from Exhibit 4.6. Provide a rationale for your score for each question.
-
Time Solutions, Inc. is an employment services firm that places both temporary and permanent workers with a variety of clients. Temporary placements account for 70% of Time Solutions' revenue;...
-
How many quarks form a baryon? A meson? What is the relationship (if any) between a quark and a lepton (e.g., an electron)?
-
What is the quark composition of the ++ baryon? It has no strangeness, no charm, and no topness or bottomness. Its spin is 3/2.
-
Distinguish between fermions and bosons in as many different ways as you can.
-
provide requirements for a ATM machine On MS word (PLEASE USE AGILE METHODOLOGY) Who is the actor on a this ATM machine What is the system? Write a User Story for the ATM machine write system...
-
A solid metal sphere with a diameter of 2 cm and a mass of 8 g is used for the following heat transfer experiments. You can assume the temperature throughout the inside of the metal sphere is...
-
Find the charters that exhibit outliers in fuel consumption per hour. Check for standardized fuel consumption per hour larger or less than -1.5 and 1.5 respectively. -- use query3 in a "from" clause.

Study smarter with the SolutionInn App


