Calculate the theoretical masses of the sucralose species in Figure 22-14 at nominal masses of 395,395, 397,397,
Question:
Calculate the theoretical masses of the sucralose species in Figure 22-14 at nominal masses of 395,395, 397,397, 399,399 and 401.401. Find the difference in ppm between observed and calculated m/z.m/z.
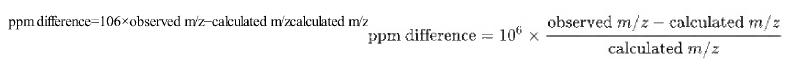
Figure 22-14

Transcribed Image Text:
ppm difference=106xobserved m/z-calculated m/zcalculated m/z ppm difference = 106 observed m/z - calculated m/z calculated m/z
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Answer Theoretical masses of the sucralose species in Figure 2214 at nominal masses of 39539...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Calculate the theoretical masses of the species in Figure 21-9 and compare your answers with the values observed in the figure. Figure 21-9 2 000 31p+ 14N16OH* 1 000 15N160* 30.97 30.98 30.99 31.00...
-
In Figure 22-14, the sucralose species with a nominal mass X=395 X=395 is [12C121H1816O835C13]-. [ 12 C 12 1 H 18 16 O 8 35 Cl 3 ]-. X+1X + 1 arises from isotopologues containing one 13C, 13 C, one...
-
The point masses m and 2m lie along the x-axis, with m at the origin and 2m at x = L. A third point mass M is moved along the x-axis. (a) At what point is the net gravitational force on M due to the...
-
How to respond to the following? Cycle stock, pipeline inventory, buffer stock, and in-transit inventory all have associated carrying costs. Interest rates on inventory values, warehousing expenses,...
-
Let the statistic U2 be as defined by Eq. (11.3.32), and let be fixed positive constant. Show that for all observed values (xi , yi), for i = 1, . . . , n, the set of points (0, 1) such that U2 < ...
-
Squirrel Tree Services reports the following amounts on December 31, 2024. In addition, the company reported the following cash flows. Required: 1. Prepare a balance sheet. 2. Prepare a statement of...
-
Define project financial management and explain its key features.
-
Turner, Inc. began work on a $7,000,000 contract in 2010 to construct an office building. During 2010, Turner, Inc. incurred costs of $1,700,000, billed their customers for $1,200,000, and collected...
-
What are the roots of the equation x^(2)-2x+2=0 in simplest a+bi form?
-
On December 1, 2020, Cambridge Printers had the account balances shown below. Debit Credit Cash 14,650 Accumulated DepreciationEquipment 1,500 Accounts Receivable 3,900 Accounts Payable 3,000...
-
Mass spectral interpretation. The compound C9H4N2Cl6 C 9 H 4 N 2 Cl 6 is a by-product found in chlorinated pesticides. a. Verify that the formula for rings + double bonds agrees with the structure....
-
When looking at the mass spectrum of an unknown substance, you tentatively identify the molecular ion MM as the peak with the most significant intensity at the high-mass end of the spectrum. Then you...
-
In the chapter opener, you learned about FastPay, a company that lends to online ad publishers based on advertising receivables. Suppose that you are running a business that relies on online ad...
-
Which method can fill in the blank that would cause the program to consistently print Tie! ten times? A. lock() B. tryLock() C. tryLock(10) D. The code does not compile regardless of what is placed...
-
Which of the following lambda expressions can be passed to a method that takes IntUnaryOperator as an argument? (Choose three.) A. v -> {System.out.print("Hello!"); return 2%1;} B. (Integer w) ->...
-
Given the table books in the figure and a ResultSet created by running the following SQL statement, which option prints the value 379? SELECT * FROM books WHERE title = 'Beginning Java' A....
-
What is the result of executing the following method? A. The declaration of name does not compile. B. The declaration of _number does not compile. C. The declaration of profit$$$ does not compile. D....
-
Which of the following could be valid JDBC URL formats for an imaginary driver named magic and a database named box? A. Only first B. Only second C. Only third D. Both first and second E. Both first...
-
Why is it difficult for union and management officials to resolve each grievance on its own merits?
-
Find the area of the surface generated by revolving the para- metric curve x = cos 1, y = sin? 1 (0 < I sa/2) about the y-axis.
-
Detection limit. A sensitive chromatographic method was developed to measure sub-part per-billion levels of the disinfectant by-products iodate (IO 3 ), chlorite (ClO 2 ), and bromate (BrO 3 ) in...
-
Olympic athletes are tested to see if they are using illegal performance-enhancing drugs. Suppose that urine samples are taken and analyzed and the rate of false positive results is 1%. Suppose also...
-
Olympic athletes are tested to see if they are using illegal performance-enhancing drugs. Suppose that urine samples are taken and analyzed and the rate of false positive results is 1%. Suppose also...
-
What is the worst case running time of the following sudo codes, in 0- notation? Suppose that all arithmetic operations (including simple multiplication) take a constant amount of time. Justify your...
-
4. Let G be a pseudorandom generator with expansion factor (n) > 2n. In each of the following cases, say whether G' is necessarily a pseudorandom generator and explain why or why not. Here, "||...
-
Write the code for the del () method in the following doubly linked list class public class ObjDList { private Obj Node list; private Obj Node tail; public ObjDList() { list = null; tail = null; }...

Study smarter with the SolutionInn App


