Mass spectral interpretation. The compound C9H4N2Cl6 C 9 H 4 N 2 Cl 6 is a by-product
Question:
Mass spectral interpretation. The compound C9H4N2Cl6 C9H4N2Cl6 is a by-product found in chlorinated pesticides.
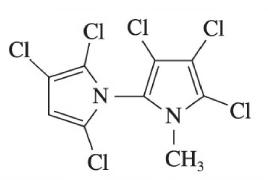
a. Verify that the formula for rings + double bonds agrees with the structure.
b. Find the nominal mass of C9H4N2Cl6.C9H4N2Cl6.
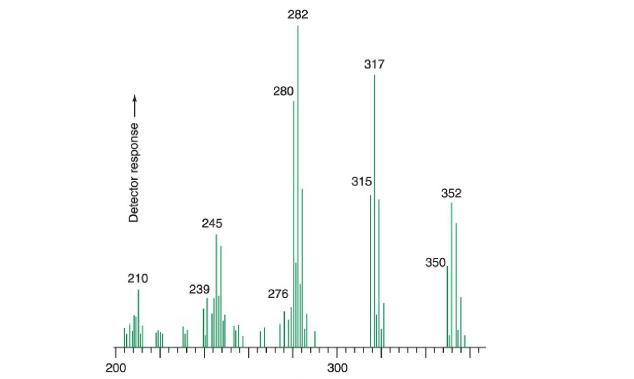
c. Suggest an assignment for m/z350,m/z350, 315,315, 280,280, 245245, and 210210 in the high-mass region of the electron ionization mass spectrum.
Transcribed Image Text:
CI Cl Cl N- CI CI N I CH3 Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a The formula for rings double bonds for the compound C9H4N2Cl6 is C9H4N...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Verify that the formula for t of Theorem 14.4 can be written as Theorem 14.4 Under the assumptions of normal regression analysis, Is a value of a random variable having the t distribution with n 2...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
Suggest a composition for the halogen compound whose mass spectrum is shown in the figure. Assign each of the major peaks. 100 127 173 160 25 158 162 7 48 HT 79 91 160 170 208 35 160 173 IITI'T1 30...
-
Consider the following graph. 2 7 6 C 10 10 H 11 E 12 15 14 (a). Find a minimum spanning tree of the graph using Kruskal's algorithm. List the edges in the order they are put into the tree. (b)....
-
For the data presented in Table 11.9, construct a confidence ellipse for β0 and β1 with confidence coefficient 0.95. Table 11.9 Data for Exercise 1 87427 00077 12100 67890...
-
Suppose a local company has the following balance sheet accounts: Calculate the missing amounts assuming the business has total assets of $37,500. Accounts Land Equipment Salaries Payable Notes...
-
Discuss project accounting and its role in managing project finances.
-
(NOL Carry back and Carry forward, Valuation Account Needed) Hayes Co. reported the following pretax financial income (loss) for the years 20092013. 2009.............................$240,000...
-
1) Explain how the different participants in the exchange market can take advantage of it. Support your argument. 20 points 2) Explain how the European Monetary Union has simplified trade in Europe....
-
The Piedmont Investment Corporation has identified four small apartment buildings in which it would like to invest. The four banks generally used by Piedmont have provided quotes on the interest...
-
Nanoparticles with a gold core and organothiol outer shell with well-defined stoichiometries Aux(SR)yAu x (SR) y i can be prepared and isolated. a. The MALDI-time-of-flight mass spectrum of the...
-
Calculate the theoretical masses of the sucralose species in Figure 22-14 at nominal masses of 395,395, 397,397, 399,399 and 401.401. Find the difference in ppm between observed and calculated...
-
What else can a business plan be used for?
-
What is the result of the following when called as java Binary.java? A. [] B. [0, 01, 1, 10] C. [0, 01, 10, 1] D. [0, 1, 01, 10] E. The code does not compile. F. The code compiles but throws an...
-
Which statements best describe the result of executing this code? (Choose two.) A. The println() causes one line of output. B. The println() causes two lines of output. C. The println() causes three...
-
What option names are equivalent to -p and -cp on the javac command? (Choose two.) A. --module-path and -classpath B. --module-path and -class-path C. --module-path and --class-path D. --path and...
-
What is the result of the following? A. Fall B. Spring C. The code does not compile. D. The code throws an exception on line s1. E. The code throws an exception on line s2. package calendar; public...
-
Which of the following can fill in the blank to print out just the number 161? import java.util.*; import java.util.stream.*; class Runner { public int getNumberMinutes() { return numberMinutes; } }...
-
Briefly discuss the broad judicial guidelines concerning unions' fair representation obligations to members. Also discuss the reasoning behind these obligations, furnishing some appropriate examples?
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
Detection limit. Low concentrations of Ni2+ -EDTA near the detection limit gave the following counts in a mass spectral measurement: 175, 104, 164, 193, 131, 189, 155, 133, 151, 176. Ten measurements...
-
What are the three parts of quality assurance? What questions are asked in each part and what actions are taken in each part?
-
Detection limit. A sensitive chromatographic method was developed to measure sub-part per-billion levels of the disinfectant by-products iodate (IO 3 ), chlorite (ClO 2 ), and bromate (BrO 3 ) in...
-
Milden Company is a merchandiser that plans to sell 4 1 , 0 0 0 units during the next quarter at a selling price of $ 5 9 per unit. The company also gathered the following cost estimates for the next...
-
How do organizational learning theories, such as the learning organization concept and knowledge management practices, contribute to sustained competitive advantage and organizational resilience in...
-
Harbert, Incorporated had a beginning balance of $12,000 in its Accounts Receivable account. The ending balance of Accounts Receivable was $10,500. During the period, Harbert collects $72,000 of its...

Study smarter with the SolutionInn App


