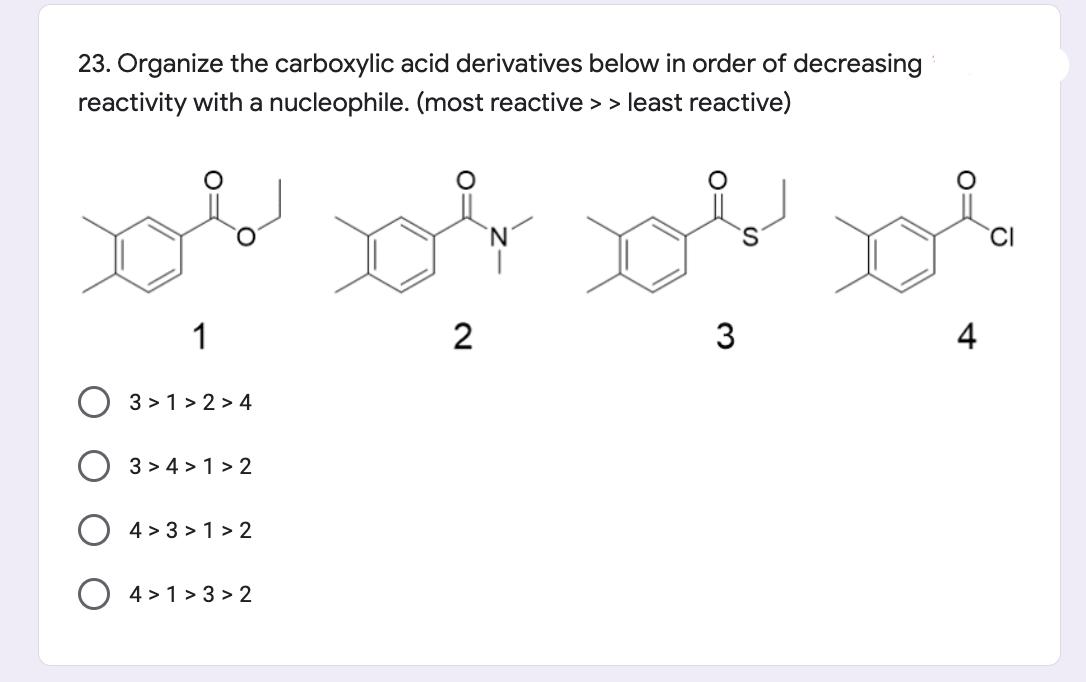
23. Organize the carboxylic acid derivatives below in order of decreasing reactivity with a nucleophile. (most...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
23. Organize the carboxylic acid derivatives below in order of decreasing reactivity with a nucleophile. (most reactive >> least reactive) CI 1 3 3 > 1 > 2 > 4 3 > 4 > 1 > 2 4 > 3 > 1 > 2 O 4 > 1 > 3 > 2 23. Organize the carboxylic acid derivatives below in order of decreasing reactivity with a nucleophile. (most reactive >> least reactive) CI 1 3 3 > 1 > 2 > 4 3 > 4 > 1 > 2 4 > 3 > 1 > 2 O 4 > 1 > 3 > 2
Expert Answer:
Answer rating: 100% (QA)
Among carboxylic acid derivatives like acid chlorides the hete... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Order these solutions in order of decreasing osmotic pressure, assuming an ideal van't Hoff factor: 0.1 M HCl, 0.1 M CaCl2, 0.05 M MgBr2, and 0.07 M Ga(C2H3O2)3
-
Rank the following compounds in order of decreasing reactivity in an E2 reaction CH3 CH3 CH3 Br Br CH3
-
List the following amides in order of decreasing reactivity toward acid-catalyzed hydrolysis: NO, CH CNH- CH CNH CH CNH NO2 CH,CNH
-
Carrefour issue its own credit card. The sales manager purchase clothes for his family for 900 using Carrefour credit card on the 1st of April. If Carrefour charge 2% per month on the amount the...
-
Use the following accounts and balances to prepare a balance sheet with the accounts in proper order for Manteno Company at June 30, 2014, using Exhibit 8 as amodel: Accounts Receivable Wages Payable...
-
Assume that Sally Smith, an interviewer, completed 50 questionnaires. Ten of the questionnaires were validated by calling the respondents and asking them one opinion question and two demographic...
-
Charles Smith, Inc., is a manufacturer of small office equipment. Smith transacts most of its business on credit and offers its customers credit terms of 2/10, n/30. On July 1, Smith shipped an order...
-
Brown Enterprises uses a word processing computer to handle its sales invoices. Lately, business has been so good that it takes an extra 3 hours per night, plus every third Saturday, to keep up with...
-
Use the Black-Scholes-Merton model to value a European put option on the spot price of a share when the strike price is $30, and the expiration is in 6 months. The current price of share is $25. The...
-
The trial balance of Rosiak Fashion Center contained the following accounts at November 30, the end of the companys fiscal year. Adjustment data: 1. Supplies on hand totaled 2,100. 2. Depreciation is...
-
(A) The following chargeable persons have Malaysian sources of income and remittances of foreign sources of income for the basis year of 2020: Chargeable persons Mr Yeo (Resident) Ms Janet...
-
You have two identical pet hamsters named Peanut Butter andJelly. The only way you can tell them apart is the custom collarthat each one wears. One day you check on Peanut Butterand Jelly and notice...
-
Compare differences between three forms of local government. Utilize the following forms to compare the differences, council-manager, strong mayor, weak mayor. Describe the authority and roles of...
-
Assume again you are interviewing for a job with a firm that you are really interested in. You have just spent 20 minutes with the HR recruiter applying the SPIN strategy in asking questions to...
-
The system in the figure is in equilibrium. A concrete block of mass 261 kg hangs from the end of the uniform strut of mass 37.9 kg. For angles = 35.6 and 0-53.7, find (a) the tension T in the cable...
-
Given the function 5x - x 5 2 a <0 f(x) = 1 Calculate the following values: f(1) = f(0) = = f(2) =
-
A 71.2-kg boy is surfing and catches a wave which gives him an initial speed of 1.60 m/s. He then drops through a height of 1.62 m, and ends with a speed of 8.51 m/s. How much nonconservative work...
-
What is your assessment of the negotiations process, given what you have studied? What are your recommendations for Mr. Reed? You must justify your conclusions
-
Which is a stronger acid? a. CH3CH2CH2OH or CH3CH==CHOH b. c. CH3CH==CHCH2OH or CH3CH==CHOH d. HCCH2OH or CH COH CH CH2CH2NH or CH3CH CHNH3
-
When the following compound is hydrated in the presence of acid, the unreacted alkene is found to have retained the deuterium atoms: What does the preceding statement tell you about the mechanism of...
-
The hydrolysis of glycinamide is catalyzed by [Co(ethylenediamine)2]2+ Propose a mechanism for this reaction. Co2+ H2NCH2CNH2 H20
-
For each of the following unrelated columns of data for the year, compute the cost of goods manufactured and the cost of goods sold: A B C Selling expenses Factory insurance. Ending finished goods...
-
Brooklyn Company reported the following amounts for October: Use Excel to prepare a schedule of cost of goods sold for October. Beginning raw materials inventory. Beginning work-in-process inventory...
-
Kelly Metal Products Corporation sold 18,000 units of product for \($40\) each during 2019. During the year, 10,000 shares of common stock were outstanding. Prepare an income statement for the year...

Study smarter with the SolutionInn App


