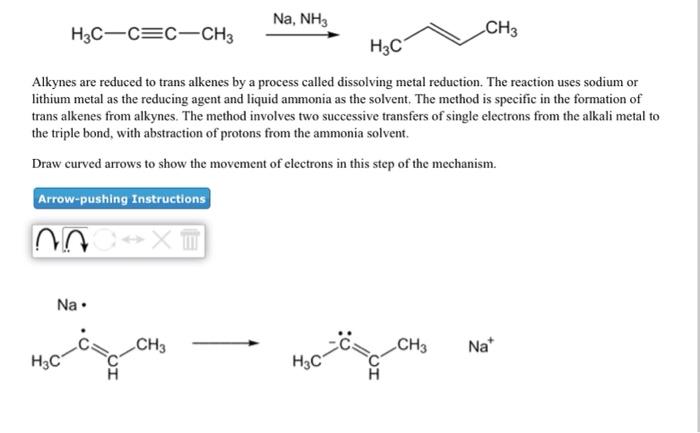
ANO XT H3C-C=C-CH3 Alkynes are reduced to trans alkenes by a process called dissolving metal reduction....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
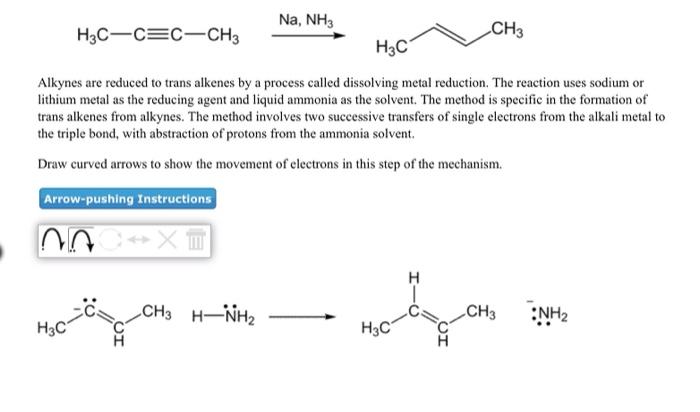
ANO XT H3C-C=C-CH3 Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Na. H3C Na, NH3 CH3 H3C H3C CH3 CH3 Na* H3C Na, NH3 H3C C C CH3 H3C Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions NOC XT CH3 HNH2 CH3 40-4₂ CH3 H3C NH₂ ANO XT H3C-C=C-CH3 Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Na. H3C Na, NH3 CH3 H3C H3C CH3 CH3 Na* H3C Na, NH3 H3C C C CH3 H3C Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions NOC XT CH3 HNH2 CH3 40-4₂ CH3 H3C NH₂
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw curved arrows to show the flow of electrons responsible for the conversion of reactants into products. a. b. c. :O CH-C OCH
-
Draw curved arrows to show the flow of electrons responsible for the conversion of the reactants into the products: Br
-
Use curved arrows to show the movement of electrons in each of the following reaction steps: a. b. c. d. Br O: " CH,COH + H-O-H CH,COH + H2O - CH3 CH3 CH CH3 CH
-
Be able to explain how changes in transportation impacted economic growth in the United States prior to WWI.
-
Do you think call center outsourcing negatively affects customer service? Explain.
-
What is the caveat the channel manager should be aware of when evaluating the performance of franchise channel members?
-
List four specific sources of financial information.
-
Warranty and Coupon Computation Schmitt Company must make computations and adjusting entries for the following independent situations at December 31, 2011. 1. Its line of amplifiers carries a 3-year...
-
Depreciation In early 2019, Sosa Enterprises purchased a new machine for $10,000 to make cork stoppers for wine bottles. The machine has a 3-year recovery period and is expected to have a salvage...
-
The manager of HerkyHornet press hires you as a consultant. She plans to order a newly-published book that is on the 2022 best-seller list. However, she is not sure about the demand for this book....
-
How can you work collaboratively with people to identify and assist them to identify positive elements of their interpersonal relationships and relationship systems? Provide at least eight examples...
-
For incompressible plane flows, what is the relationship between flow function and vorticity?
-
Under what circumstances can the viscosity of the fluid be reflected? From the perspective of the transport properties of fluid movement, what kind of transport characteristics does fluid viscosity...
-
Prove that equipotential lines and streamlines are orthogonal to each other.
-
Write down the deformation rate matrix of fluid clusters and its three invariants? Explain the physical meaning of divergence?
-
Please explain the physical meaning of Newtons internal friction law? What is the physical mechanism of the dynamic viscosity coefficient?
-
the following five psychological research methods for gathering information about human behavior are discussed: (1) naturalistic observation, (2) case studies, (3) surveys, (4) correlational studies,...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Several additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitro compounds and the Gabriel synthesis leave the carbon chain unchanged. Formation...
-
Draw and name all the chlorinated benzenes having from one to six chlorine atoms.
-
Propose structures for intermediates and products (A) through (K). Mg, ether (1) CH CH2CHO (2) H,O l-bromobutane (D) KOH. H2O HzSO, heat (D O3 (2) (CH3.S CH,CH CH CHO + CH3(CH2CHO CH,CH,Br (1) NaNH2,...
-
Sketch (by hand) the graphs of each of the following functions over four periods. Then sketch the extensions of each of the functions as both an even and odd periodic function. Determine the...
-
Consider the function \(f(x)=x,-\pi
-
Differentiate the Fourier sine series term by term in Problem 18. Show that the result is not true. Why not? Data from Problem 18 Consider the function \(f(x)=x, 0

Study smarter with the SolutionInn App


