NADH and FADH2 are produced by the Citric Acid Cycle (CAC) and these electron shuttles go...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
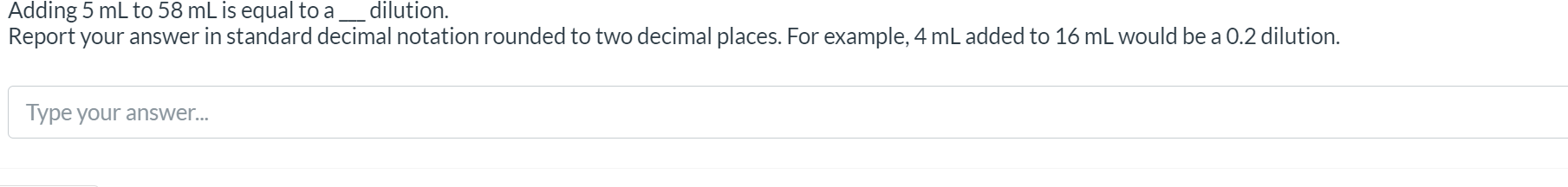
NADH and FADH2 are produced by the Citric Acid Cycle (CAC) and these electron shuttles go on to the Electron Transport Chain (ETC). A mole of NADH will produce 3 moles of ATP from the ETC. A mole of FADH₂ will produce 2 moles of ATP from the electron transport chain. A single mole of pyruvate will produce 4 moles of NADH and 1 mole of FADH₂ from the CAC. Given: The ETC produced 99 moles of ATP from the products of the CAC. Find: How many moles of pyruvate entered the CAC? Note: Brock, a Biology of Microorganisms considers the synthesis of Acetyl CoA from Pyruvate to be part of the Citric Acid Cycle. This is different than what you may have learned in BIOL-151 where the Morris Book considers Acetyl Co-A Synthesis a separate step. See page 89 of Brock for full details. Round your answer to one decimal place. IMPORTANT NOTE ABOUT ANSWER FORMAT Report your answer in standard decimal notation rounded to 2 decimal places. o 5.0125 moles of pyruvate would be reported as 5.01 O 5.01*10^0, or 0.50*10^1, or 5.0125 would be marked wrong. ● • If your answer is a whole number you must still include the trailing zeros!! o e.g. 17 pyruvates must be reported as 17.00 to get credit o likewise, 17.1 must be reported as 17.10 to get credit Neisseriales is part of which subgroup of proteobacteria? O Alphaproteobacteria O Betaproteobacteria Gammaproteobacteria O Deltaproteobacteria O Epsilonproteobacteria Adding Report 5 mL to 58 mL is equal to a ____ dilution. your answer in standard decimal notation rounded to two decimal places. For example, 4 mL added to 16 mL would be a 0.2 dilution. Type your answer... NADH and FADH2 are produced by the Citric Acid Cycle (CAC) and these electron shuttles go on to the Electron Transport Chain (ETC). A mole of NADH will produce 3 moles of ATP from the ETC. A mole of FADH₂ will produce 2 moles of ATP from the electron transport chain. A single mole of pyruvate will produce 4 moles of NADH and 1 mole of FADH₂ from the CAC. Given: The ETC produced 99 moles of ATP from the products of the CAC. Find: How many moles of pyruvate entered the CAC? Note: Brock, a Biology of Microorganisms considers the synthesis of Acetyl CoA from Pyruvate to be part of the Citric Acid Cycle. This is different than what you may have learned in BIOL-151 where the Morris Book considers Acetyl Co-A Synthesis a separate step. See page 89 of Brock for full details. Round your answer to one decimal place. IMPORTANT NOTE ABOUT ANSWER FORMAT Report your answer in standard decimal notation rounded to 2 decimal places. o 5.0125 moles of pyruvate would be reported as 5.01 O 5.01*10^0, or 0.50*10^1, or 5.0125 would be marked wrong. ● • If your answer is a whole number you must still include the trailing zeros!! o e.g. 17 pyruvates must be reported as 17.00 to get credit o likewise, 17.1 must be reported as 17.10 to get credit Neisseriales is part of which subgroup of proteobacteria? O Alphaproteobacteria O Betaproteobacteria Gammaproteobacteria O Deltaproteobacteria O Epsilonproteobacteria Adding Report 5 mL to 58 mL is equal to a ____ dilution. your answer in standard decimal notation rounded to two decimal places. For example, 4 mL added to 16 mL would be a 0.2 dilution. Type your answer...
Expert Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these biology questions
-
The first step in the citric acid cycle is reaction of oxaloacetate with acetyl CoA to give citrate. Propose a mechanism, using acid or base catalysis asneeded. "O2C. 02C H .co2 CO SCOA Citrate...
-
The citric acid cycle is a series of biological reactions that plays a central role in cell metabolism. The cycle includes dehydration reactions of both malic and citric acids, yielding fumaric and...
-
The citric acid cycle can be divided into two phases with respect to the oxidation of acetyl- CoA. Describe each phase and write its balanced equation.
-
1. Consider a local department store which only sells jeans (J) and sweaters (S). Jeans cost $20 and sweaters cost $30. For each of the examples below, graph the budget constraint our consumer faces,...
-
Assuming the pure expectations theory is correct, an upward-sloping yield curve implies: a. Interest rates are expected to increase in the future. b. Longer-term bonds are riskier than short-term...
-
Evaluate the following humorous analogy between the murder of a famous Roman emperor and the deadening effect of meetings: This month is the 2,053rd anniversary of the death of Julius Caesar, who...
-
Several 95% confidence intervals for the mean response will be constructed, based on a data set for which the sample mean value for the explanatory variable is x = 10. The values of x for which the...
-
Read Appendix 9-1. Referring to Exhibit 9-14 and related notes, assume instead that Toyozas inventories were costed using the FIFO method and that Lincoln Enterprises employed the LIFO method....
-
A client maintains a large data center where accesd is limited to authorized employees. How may an auditor best determine the effectiveness of this control activity?
-
Integer arrays originalArr and cleaning Arr are read from input, each containing three elements. If an element in cleaningArr is equal to 1, replace the corresponding element in original Arr with O....
-
A. In a series of semi-annual payments of P14465 each, the first payment is due at the beginning of 5 years and the last at the end of 12 years and 6 months. If money is worth 11% compounded...
-
What is the current total product cost (for the 20 million units) including fixed and variable cost? Great-Cola spends $3 on direct materials, direct labor, and variable manufacturing overhead for...
-
Determine the moment of force of240 lb regarding: a) To point A (climb) b) At the linnea DB(vectorial)
-
Superior Micro Products uses the weighted - average method in its process costing system. Data for the Assembly Department for May appear below: Materials Labor Overhead Work in process, May 1 $ 1 9...
-
Blocks A (mass 7.00 kgkg) and B (mass 12.00 kgkg, to the right of A) move on a frictionless, horizontal surface. Initially, block B is moving to the left at 0.500 m/sm/s and block A is moving to the...
-
During October 2 0 2 3 , Fern Field Farms, Inc. received $ 1 0 , 0 0 0 from customers in exchange for fruit and vegetables. During the same month, the company paid $ 2 , 0 0 0 to employees, $ 5 0 0...
-
Q3. Consider a bar of cross-section A and length L in fig.1. A uniform tensile load P is applied Tzx, Txy) and strain (&, ) to the two ends of the rod; find the state of stress (x, Ey, E, Vyz, Vzx,...
-
Explain how two samples can have the same mean but different standard deviations. Draw a bar graph that shows the two samples, their means an standard deviations as error bars. T S
-
Deduce structures for each of the following. (a) Molecular formula C 6 H 10 ; NMR spectrum A; no strong IR bands between 2100 and 2300 or 3250 and 3350 cm -1 . (b) Molecular formula C 7 H 12 ; NMR...
-
Reaction review. Without consulting the Reaction Road Map on p. 721, suggest a reagent to convert benzene into each of the following compounds. C(CH3)3 NO2 CH3 CH,CH; SO,H Br (e) (f) (h)
-
(a) Sketch a potential energy/reaction coordinate graph showing the two propagation steps for the monobromination of pentane to give the major product (Problem 30). Use DH information from this...
-
Use Lagrange's equation to derive the equation of motion for the simple pendulum of Example 4.15, except here assume that the mass \(m\) is suspended on a rigid bar that is connected to the support...
-
Resolve Problem 19 including damping. Discuss the importance of damping by comparing the two results. Problem 19: Structures are sometimes subjected to very rapidly applied loads of extremely short...
-
Solve for the response for all time of an underdamped oscillator that is driven by the forcing function shown in (a) Figure 4.34, and (b) Figure 4.35. Use the convolution integral. -2.0 -1.5 -1.0 1...

Study smarter with the SolutionInn App



