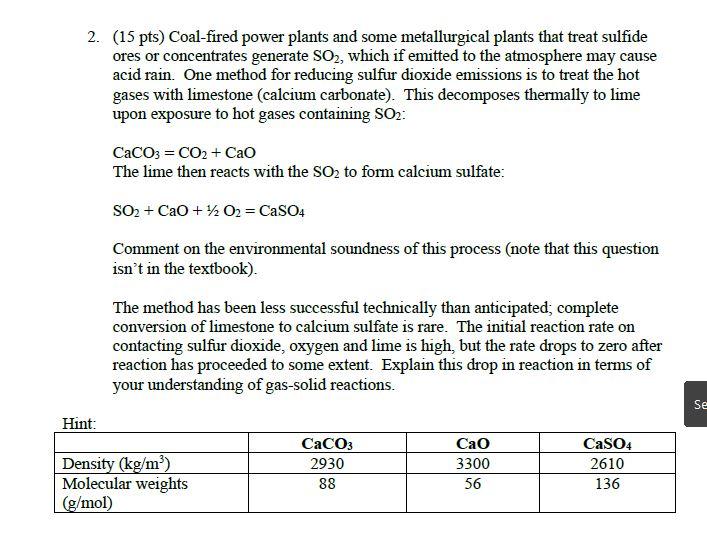
2. (15 pts) Coal-fired power plants and some metallurgical plants that treat sulfide ores or concentrates...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. (15 pts) Coal-fired power plants and some metallurgical plants that treat sulfide ores or concentrates generate SO2, which if emitted to the atmosphere may cause acid rain. One method for reducing sulfur dioxide emissions is to treat the hot gases with limestone (calcium carbonate). This decomposes thermally to lime upon exposure to hot gases containing SO₂: Hint: CaCO3 = CO2 + CaO The lime then reacts with the SO₂ to form calcium sulfate: SO₂ + CaO + 1/2O2 = CaSO4 Comment on the environmental soundness of this process (note that this question isn't in the textbook). The method has been less successful technically than anticipated; complete conversion of limestone to calcium sulfate is rare. The initial reaction rate on contacting sulfur dioxide, oxygen and lime is high, but the rate drops to zero after reaction has proceeded to some extent. Explain this drop in reaction in terms of your understanding of gas-solid reactions. Density (kg/m³) Molecular weights (g/mol) CaCO3 2930 88 CaO 3300 56 CaSO4 2610 136 Se 2. (15 pts) Coal-fired power plants and some metallurgical plants that treat sulfide ores or concentrates generate SO2, which if emitted to the atmosphere may cause acid rain. One method for reducing sulfur dioxide emissions is to treat the hot gases with limestone (calcium carbonate). This decomposes thermally to lime upon exposure to hot gases containing SO₂: Hint: CaCO3 = CO2 + CaO The lime then reacts with the SO₂ to form calcium sulfate: SO₂ + CaO + 1/2O2 = CaSO4 Comment on the environmental soundness of this process (note that this question isn't in the textbook). The method has been less successful technically than anticipated; complete conversion of limestone to calcium sulfate is rare. The initial reaction rate on contacting sulfur dioxide, oxygen and lime is high, but the rate drops to zero after reaction has proceeded to some extent. Explain this drop in reaction in terms of your understanding of gas-solid reactions. Density (kg/m³) Molecular weights (g/mol) CaCO3 2930 88 CaO 3300 56 CaSO4 2610 136 Se
Expert Answer:
Answer rating: 100% (QA)
2 CCO CO The lime then reacts with the so to from calcium suffate 50 CO 10 C 504 process The a... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
You have exams in economics and chemistry coming up, and you have 5 hours available for studying. The following table shows the trade-offs you face in allocating the time you will spend studying each...
-
What stereo chemistry do you expect for the tri-substituted alkene obtained by E2 elimination of the following alkyl halide on treatment with KOH? (Reddish brown =Br.)
-
Name the following amine, including R, S stereo chemistry and draw the product of its reaction with excess iodomethane followed by heating with Ag2O (Hofmann elimination). Is the stereochemistry of...
-
Here are the supply and demand equations for throstles, where p is the price in dollars: D (p) = 40 p S (p) = 10+p. On the axes below, draw the demand and supply curves for throstles, using blue...
-
AutoCare Ltd. (ACL) is a federally incorporated public company formed in 2007 to manufacture and sell specialty auto products such as paint protection and rust proofing. By 2010, ACL's board of...
-
Calculate the fraction of the conduction electrons in tungsten \(\left(\varepsilon_{F}=9.0 \mathrm{eV}ight)\) at \(3000 \mathrm{~K}\) whose kinetic energy \(\varepsilon\left(=\frac{1}{2} m...
-
A Boeing 747 aircraft weighing \(580,000 \mathrm{lb}\) when loaded with fuel and 100 passengers takes off with an airspeed of \(140 \mathrm{mph}\). With the same configuration (i.e., angle of attack,...
-
Odd Wallow Drinks is considering adding a new line of fruit juices to its merchandise products. This line of juices has the following prices and costs: Selling price per case (24 bottles) of juice ....
-
3. Starting a Brazilian Ju Jitsu (BJJ) studio Lisa has practiced BJJ over the past 2 years and although she is only a blue belt, she thinks she knows enough to turn her hobby into a business. While...
-
December 31, 2014, trial balances for Pledge Company and its subsidiary Stom Company follow: Pledge Company purchased 72,000 shares of Stom Company's common stock on January 1, 2011, for $300,000. On...
-
A company needs to choose from four potential projects to undertake, where each project would run for 3 years and have the following characteristics: Project Return (Em) 1 2 3 0.2 0.3 0.5 0.1 4...
-
An architect has designed two tunnels. Tunnel A is modeled by x + y + 28x + 52 = 0, and tunnel B is modeled by x - 36x + 16y + 68 = 0, where all measurements are in feet. The architect wants to...
-
A scientific experiment consists of a sequence of 12 tests. Each trial of a test has a 30% chance of passing, and a successful attempt of a test is required to move on to the next test. The success...
-
In September 2020, Ayayai Corp. commits to selling 149 of its iPhone-compatible docking stations to Better Buy Co. for $16,390 ($110 per product). The stations are delivered to Better Buy over the...
-
Determine the derivative of each function. (a) y = x2x +5 (2x-6)4 (x+2)3 (b) y: 8. [6 marks]. A tangent to the parabola y = x - 4x + 5 is perpendicular to 2x + 8y + 50. Determine the equation of the...
-
1. 2. Find (x) if f(x)=x. 3. 4. If f(x)=In(x+4+e"), then what is f'(0) ? 5. Find () (3) for f(x)=x +5x-3. (E+xalx 61 If the slope of a strictly monotonic function, f. is slope off at the point (q....
-
Using Johnson and Johnson Company, pick one material issue they are currently facing and see if the company has a sustainability goal for this issue. Is it ASMART? Please provide a detailed...
-
Could a set of three vectors in span all of? Explain. What about n vectors in when n is less than m? R4
-
Effectiveness of drug tests of Olympic athletes. Erythropoietin (EPO) is a banned drug used by athletes to increase the oxygen-carrying capacity of their blood. New tests for EPO were first...
-
MINITAB was used to fit the complete second-order model E(y) = β0 + β1x1 + β2x2 + β3x1x2 + β4x12 + β5x22 To n = 39 data points....
-
Suppose x is a normally distributed random variable with = 30 and = 8. Find a value x0 of the random variable x such that a. P(x x0) = .5 b. P(x < x0) = .025 c. P(x > x0) = .10 d. P(x > x0) = .95
-
Someone who exposes the ethical misdeeds of others in an organization is usually called a/an _________. (a) whistleblower (b) ethics advocate (c) ombudsman (d) stakeholder
-
If a manager fails to enforce a late-to-work policy for all workersthat is, by allowing some favored employees to arrive late without penaltiesthis would be considered a violation of _________. (a)...
-
According to research on ethics in the workplace, _________ is/are often a major and frequent source of pressures that create ethical dilemmas for people in their jobs. (a) declining morals in...

Study smarter with the SolutionInn App


