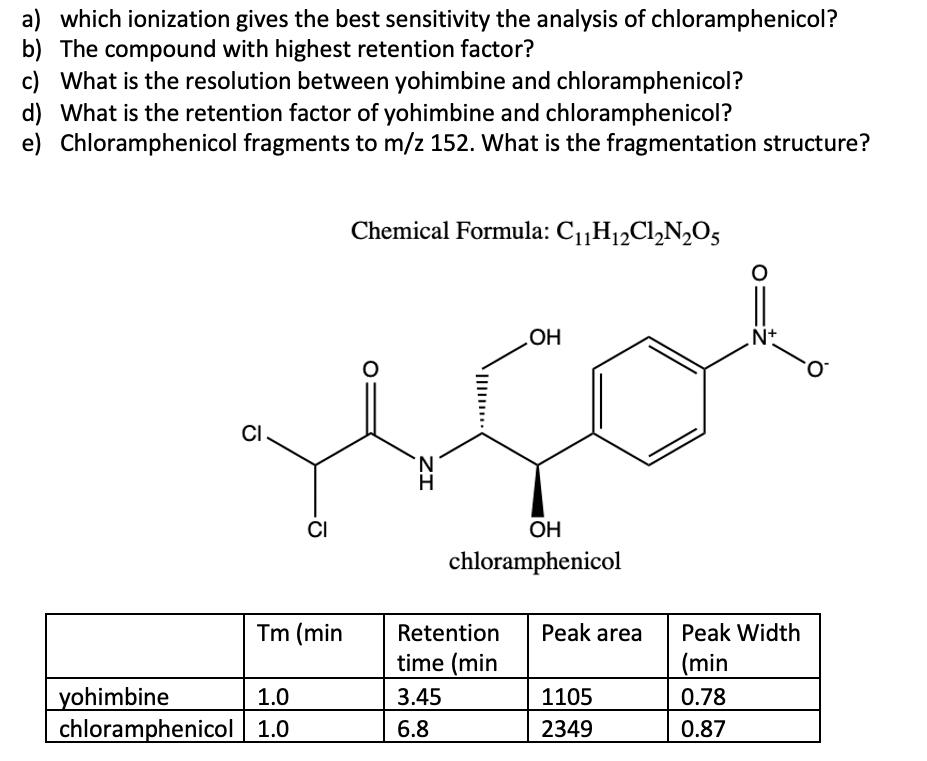
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI
Expert Answer:
Answer rating: 100% (QA)
a The electrospray negative ionization Electrospray Ionization ESI probe in negative ionization mode ... View the full answer

Related Book For 

Applied Statistics in Business and Economics
ISBN: 978-0073521480
4th edition
Authors: David Doane, Lori Seward
Posted Date:
Students also viewed these chemistry questions
-
Compound C (C9H11NO) gives a positive Tollens' test (can be oxidized to a carboxylic acid) and is soluble in dilute HCl. The IR spectrum of C shows a strong band near 1695 cm-1 but shows no bands in...
-
A compound with molecular formula C4H6O gives the infrared spectrum shown in Figure 13.34. Identify the compound. 3.5 4 5 12 13
-
compound with molecular formula C5H10O2 gives the following IR spectrum. When it undergoes acid-catalyzed hydrolysis, the compound with the following 1H NMR spectrum is formed. Identify the...
-
Prove the following statement: If a transformer having a series impedance Ze is connected as an autotransformer, its per-unit series impedance Z as an autotransformer will be SE Zoq NSE + NC Note...
-
In the previous problem, assume the equity increases by 1,100 solaris due to retained earnings. If the exchange rate at the end of the year is 1.24 solaris per dollar, what does the balance sheet...
-
Newton's rings can be seen when a planoconvex lens is placed on a flat glass surface (see Problem 3539). For a particular lens with an index of refraction of n = 1.50 and a glass plate with an index...
-
In a lawsuit against Angies List, Inc. for overtime pay, sales representatives were paid based on a 40-hour workweek but alleged that they routinely worked 50 to 60 hours a week without extra...
-
A list of 16 issues that led to incorrect formulations in Richard Dulskis jam manufacturing unit is provided below: 1. Incorrect measurement 2. Antiquated scales 3. Lack of clear instructions 4....
-
All work must be shown on every facet of the solution. This includes a timeline with cashflows, timing and rates clearly laid out and formulas (in pure form, not in the calculator or excel form). A...
-
A columnist made the following comment in an online article: "In any case, the Q-factor approach, unlike consumption CAPM, explains where the anomalies come from (and why they might end). Consumption...
-
Company defaulted on a $160,000 loan that was due on December 31, 2024. The bank has agreed to allow Lowlife to repay the $160,000 by making a series of equal annual payments beginning on December...
-
Under IAS 10 Events After the Reporting Period, there are two categories of subsequent events. ABC Inc. is preparing its financial statements for the year ended October 31, Year 1. What is one of the...
-
You are given a dataset. The dataset file name is CAR_ACCIDENTS_VEECTORIA.csv. The file size is 2 kb. The data was created on 2 Jan 2020. The dataset was provided and prepared by Veectorian...
-
Red, White and Blue are equal partners. White retires, his share of goodwill is ~ 18,000. The remaining partners have decided to continue the business sharing profits and losses in the ratio of 3 :...
-
A professor was interested in comparing how well her students performed on their midterm and final exams in her calculus course. She randomly sampled 7 students from her class and recorded their...
-
Should I deduct capital dividend received from investment from net income for tax purpose?
-
Consider the workpiece shown in the figure below, where three holes are to be drilled at different places. The depth of hole is equal 10mm, Z= 0mm at the surface of the workpiece, the cutting tool is...
-
An access route is being constructed across a field (Figure Q8). Apart from a relatively firm strip of ground alongside the field's longer side AB, the ground is generally marshy. The route can...
-
Engineers are testing company fleet vehicle fuel economy (miles per gallon) performance by using different types of fuel. One vehicle of each size is tested. Does this sample provide sufficient...
-
At a University of Colorado woman's home basketball game, a random sample of 25 con- cession purchases showed a mean of $7.12 with a standard deviation of $2.14. For the next week's home game, the...
-
A manufacturing project has five independent phases whose completion must be sequential. The time to complete each phase is a random variable. The mean and standard deviation of the time for each...
-
Recall the facts in Leonard v. Pepsico Had this been a contract, would it have been subject to the Statute of Frauds?
-
Online purchases are commonly governed by a sales contract between the online merchant and the consumer in the terms of use found as a link on the sellers home page. Often, purchasers are informed...
-
Zapata, a Texas company, entered into a contract for Unterweser, a German company, to tow an oil-drilling rig from Louisiana to Italy. The contract stated, Any dispute arising must be treated before...

Study smarter with the SolutionInn App


