Consider the Cu-Ag phase diagram (Figure 9.7 p. 298). At a temperature of 900 degree C...
Fantastic news! We've Found the answer you've been seeking!
Question:
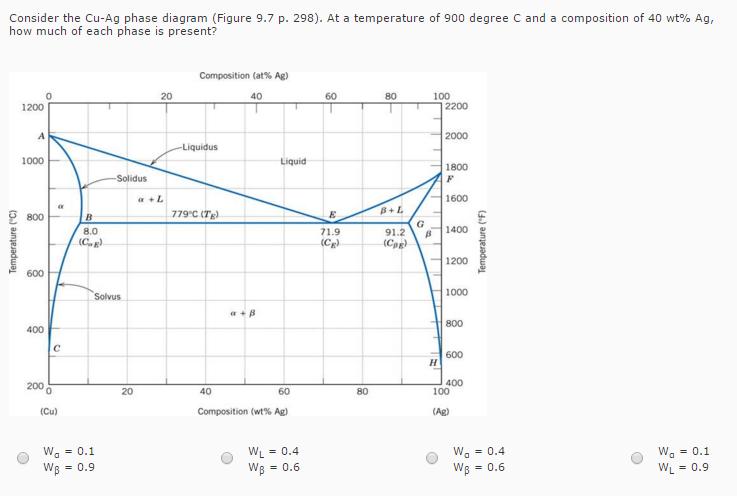
Transcribed Image Text:
Consider the Cu-Ag phase diagram (Figure 9.7 p. 298). At a temperature of 900 degree C and a composition of 40 wt% Ag, how much of each phase is present? Composition (at% Ag) 20 40 60 80 100 1200 2200 A 2000 -Liquidus 1000 Liquid 1800 Solidus 1600 B+L 800 B 779°C (Tg) 8.0 1400 71.9 (Cg) 91.2 (Cg) (Cog) 1200 600 1000 Solvus 800 400 600 H 200 0 400 100 20 40 60 80 (Cu) Composition (wt% Ag) (Ag) W. = 0.1 We = 0.9 WL = 0.4 W. = 0.4 W. = 0.1 WB = 0.6 Wg = 0.6 WL = 0.9 Temperature ("C) Temperature ("F) Consider the Cu-Ag phase diagram (Figure 9.7 p. 298). At a temperature of 900 degree C and a composition of 40 wt% Ag, how much of each phase is present? Composition (at% Ag) 20 40 60 80 100 1200 2200 A 2000 -Liquidus 1000 Liquid 1800 Solidus 1600 B+L 800 B 779°C (Tg) 8.0 1400 71.9 (Cg) 91.2 (Cg) (Cog) 1200 600 1000 Solvus 800 400 600 H 200 0 400 100 20 40 60 80 (Cu) Composition (wt% Ag) (Ag) W. = 0.1 We = 0.9 WL = 0.4 W. = 0.4 W. = 0.1 WB = 0.6 Wg = 0.6 WL = 0.9 Temperature ("C) Temperature ("F)
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these mechanical engineering questions
-
Consider a liquid geothermal resource at a temperature of 110oC. An investor is considering a power plant construction on this site, and the investor is asking for your opinion whether this is an...
-
Consider a system in thermal equilibrium at a temperature of 30 K. 1 x 10-7 J of heat is added to the system. Calculate by what factor the number of accessible states of the system will increase....
-
A large body of nonluminous gas at a temperature of 1100?C has emission bands between 2.5 and 3.5 mm and between 5 and 8 mm. At 1100?C, the effective emittance in the first band is 0.8 and in the...
-
Using algebra, find the slope and y-intercept of the line represented by each of the following equations. (a) 7x + 3y = 6 (b) 10y = 5x (c) (2y 3x)/2 = 4 (d) 1.8x + 0.3y 3 = 0 (e) 1/3x = -2 (f) 11x...
-
What is the distinction between a sale and an agreement to sell? What is the significance of that distinction?
-
Use the unit step function to find an equation for each graph in terms of the function y = f (t), whose graph is given in Figure 7.R.1. y=f(t) to
-
Marion Manufacturing, a steel company, announces that it will be taking a major restructuring charge that will lower earnings this year by $500 million. Assume that the charge is not tax deductible...
-
Relevant-cost approach to pricing decisions. Burst, Inc., cans peaches for sale to food distributors. All costs are classified as either manufacturing or marketing. Burst prepares monthly budgets....
-
use links. 'ou have just been hired by Intel in its ?nance division. Your first assignment is to deter- mine the net cash flows and NPV of a proposed new generation of mobile chips. Capital...
-
You are employed by McDowell and Partners, Chartered Accountants (M&P). A new client, Community Finance Corporation (CFC), approached M&P for assistance. Enviro Ltd. (Enviro) has asked CFC for a loan...
-
Simon creates a trust with income to Alvin for Alvin's life, then income to Simon for Simon's life, and a remainder to Brittany or Brittany's estate. Simon dies, predeceasing all other parties. What...
-
I am a professional accountant and hold the position of financial analyst, capital projects, with the Town of Pinecrest. In my position, I deal with, among others, developers and their lawyers with...
-
Based on customer demand and competition, an auto manufacturer has decided to offer a 100,000-kilometer warranty on all parts. Using current parts and production standards, the warranty will cost the...
-
I have a question that I need a bit of help on, but I am not sure where to turn, and I hope you may be able to help me out. I am the CFO of a charitable organization, it is a paid position and I am a...
-
Precision Timepieces has identified customers who would be willing to buy stopwatches with specific characteristics for 20 apiece. Precision wants a 20 percent profit margin on anything that it makes...
-
I am the assistant controller at a mediumsized, not-for-profit organization. I hired a new accounts-payable clerk three months agolets call her Mary, which is not her real nameand then I fired her...
-
Windswept, Incorporated2021 Income Statement($ in millions)Net sales$ 9,200Cost of goods sold7,550Depreciation430Earnings before interest and taxes$ 1,220Interest paid92Ta 2 answers
-
1. True or False. Pitfalls to consider in a statistical test include nonrandom samples, small sample size, and lack of causal links. 2. Because 25 percent of the students in my morning statistics...
-
Consider gas flow through a converging nozzle with specified inlet conditions. We know that the highest velocity the fluid can have at the nozzle exit is the sonic velocity, at which point the mass...
-
Air is heated as it flows through a 6 in 6 in square duct with negligible friction. At the inlet, air is at T1 = 700 R, P1 = 80 psia, and V1 = 260 ft/s. Determine the rate at which heat must be...
-
Consider two cars with one using ethanol and the other using biodiesel as the fuel. For a given full tank of liquid fuel, which car gets more mileage? Why?
-
Question: Sports Graphics, Inc. imports "Chill" brand coolers from Taiwan. Chill coolers have an outer shell of vinyl, with handles and pockets, and an inner layer of insulation. In a recent lawsuit,...
-
Question: In a recent WTO case, several nations claimed that American laws concerning shrimp fishing were unfair and illegal. The case demonstrated a conflict between two important values. What were...
-
Question: Flyby Knight (FK) contracts to sell 12 helicopters to Air Nigeria, for $8 million each. Payment is to be made by letter of credit, issued by the Bank of Nigeria, confirmed by Citibank in...

Study smarter with the SolutionInn App


