Estimate the value of Los Pollos Hermanos' equity by treating the levered equity like a call...
Fantastic news! We've Found the answer you've been seeking!
Question:
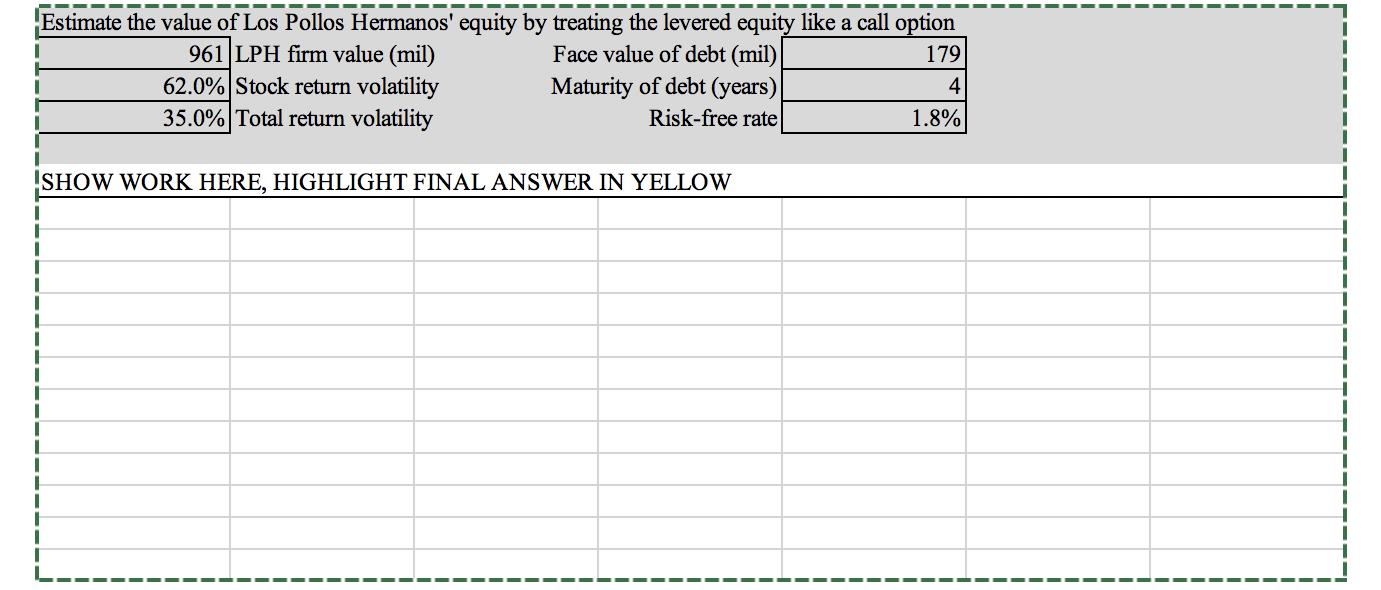
Transcribed Image Text:
Estimate the value of Los Pollos Hermanos' equity by treating the levered equity like a call option 961 LPH firm value (mil) 62.0% Stock return volatility 35.0% Total return volatility Face value of debt (mil) 179 Maturity of debt (years) Risk-free rate 4 1.8% SHOW WORK HERE, HIGHLIGHT FINAL ANSWER IN YELLOW Estimate the value of Los Pollos Hermanos' equity by treating the levered equity like a call option 961 LPH firm value (mil) 62.0% Stock return volatility 35.0% Total return volatility Face value of debt (mil) 179 Maturity of debt (years) Risk-free rate 4 1.8% SHOW WORK HERE, HIGHLIGHT FINAL ANSWER IN YELLOW
Expert Answer:
Answer rating: 100% (QA)
The value of Los Pollos Hermanos equity can be estimated by treating the levered equity like a call ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Estimate the value of y= Cp/Cv for carbon dioxide. Do this calculation with and without the vibrational contribution to the energy. Which is closer to the expected experimental value at 25C?
-
Estimate the value of the autocorrelation Corr (t, t 1) if the Durbin-Watson statistic (a) D = 0.8? (b) D = 1.5? (c) D = 3?
-
A venture investor wants to estimate the value of a venture. The venture is not expected to produce any free cash flows until the end of Year 6, when the cash flow is estimated at $2,000,000, and is...
-
2.) A truck with axle loads of W = 19.6kN and W2 = 78.6kN on a wheel base of d = 4.3m rolls across the beam shown in the figure. Determine the following: 3m 25 m 22 m a.) Draw the influence line for...
-
Beauty Division Store has a new promotional program that offers a free gift-wrapping service for its customers. Beautys customer service division has practical capacity to wrap 7,500 gifts at a...
-
Pretty Pets is a small etail business specializing in the sale of exotic pet gifts and accessories over the web. The business is owned by a sole proprietor and operated out of her home. Results for...
-
You are a salesperson working for American Airlines calling on the vice president of a large manufacturing company. Many of the companys people travel all over the world, and you would like them to...
-
Rembrandt Frame Company prepared the following sales budget for the current year: At the end of December 2008, the following unit sales data were reported for the year: For the year ending December...
-
Discuss what is meant by a marketspace, its components and participants, and types. Are there winners and losers in the e - marketplace? Why, or why not? Please discuss and use real life examples to...
-
Other information: Intercompany sales: On January 1, Year 10: Salt had on hand $30,000 of inventory purchased from Pillar. Pillar had on hand $90,000 of inventory purchased from Salt. Both companies...
-
1. Suppose that four companies, Alpha, Beta, Gama, and Delta, own the shares of the Epsilon company. Alfa owns 49% of the common shares of psilon. Beta, Gamut and Delta have 17% each. The psilon...
-
Why is it important to first forecast the projects local currency cash flows?
-
How can a company alter its translation exposure?
-
What does it mean to describe financing as a global procurement decision?
-
When evaluating foreign projects, why is discounting residual cash flows to equity holders at the equity cost of capital preferable to discounting free cash flows at the weighted cost of capital?
-
What is the true cost of translation exposure hedging?
-
Draw temperature curves dry air parcels that we release with temperatures of 22C and 24C at the ground. The y-curve in the graph describes the environmental temperature profile. At what altitudes do...
-
Danielle has an insurance policy with a premium of $75 per month. In September she is in an accident and receives a bill worth $2990 for the repair of her own property. Her deductible is $250 and her...
-
M.-G. Olivier and R. Jadot Cl. Chem. Eng. Data 42,230 (1997)) studied the adsorption of butane on silica gel. They report the following amounts of absorption (in moles per kilogram of silica gel) at...
-
Calculate the change in Gibbs energyof25 g of methanol (mass density 0.791 g cm-3) when the pressure is increased isothermally from 100 kPa to 100 MPa.
-
From data in Table 18.1 calculate the molar polarization, relative permittivity, and refractive index of methanol at 20e. Its density at that temperature is 0.7914 g cm-1,
-
Describe what each letter stands for in the CVP graph. D C 300 200 F G H B A 150 E The breakeven point is at K units and at dollars of sales.
-
For each of the situations listed, identify the primary standard from the IMA Statement of Ethical Professional Practice that is violated (competence, confidentiality, integrity, or credibility). 1....
-
Refer to your answer to S7-7. a. Compute the total number of dinner cruises that FunTime Cruiseline must sell to break even. b. Compute the number of regular cruises and executive cruises the company...

Study smarter with the SolutionInn App


