FeS can be roasted in 02 to form FeO: 2FeS +302 2FeO +2502 If the...
Fantastic news! We've Found the answer you've been seeking!
Question:
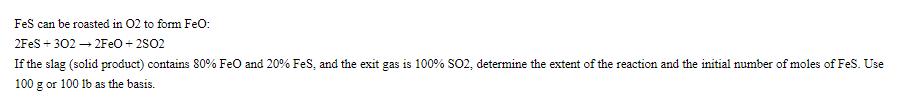 Â
Â
Transcribed Image Text:
FeS can be roasted in 02 to form FeO: 2FeS +302 → 2FeO +2502 If the slag (solid product) contains 80% FeO and 20% FeS, and the exit gas is 100% SO2, determine the extent of the reaction and the initial number of moles of FeS. Use 100 g or 100 lb as the basis. FeS can be roasted in 02 to form FeO: 2FeS +302 → 2FeO +2502 If the slag (solid product) contains 80% FeO and 20% FeS, and the exit gas is 100% SO2, determine the extent of the reaction and the initial number of moles of FeS. Use 100 g or 100 lb as the basis.
Expert Answer:
Answer rating: 100% (QA)
Calculate the molar masses of FeS IronII Sulfide FeO IronII Oxide and SO2 Sulfur Dioxide FeS Fe 5585 ... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these physics questions
-
DreamBuild Construction Company has been in business for many years. His accountant has extracted the following trial balance from his books of account as at 3 1 March 2 0 2 2 : OMR OMR Debtor...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Jay Bhattacharya and Kate Bundorf of Stanford University have found evidence that people who are obese and who work for firms that provide health insurance receive lower wages than workers at those...
-
What can happen if you cosign a loan?
-
Are there any disadvantages to be a first-mover. Provide examples.
-
The bank columns in the cash book for June 19X7 and the bank statement for that month for C Grant are as follows: You are required to: (a) Write the cash book up to date to take the above into...
-
Propane gas enters a continuous adiabatic heat exchanger? at 40?C and 250 kPa and exits at 240?C superheated steam at 300?C and 5.0 bar enters the exchanger flowing counter currently to the propane...
-
Bill plans to open a self-serve grooming center in a storefront.The grooming equipment will cost $435,000. He expects aftertax cashinflows of $94,000 annually for eight years, after which he planst 2...
-
Senior Home Living (SHL) is a Canadian-based corporation located in British Columbia. SHL provides senior living residences across Canada. The company was incorporated in 1975, and has been...
-
Given Standard and Modified Proctor compaction test results for a soil. The contract specifications require a dry density of at least 113 pcf and a water content of 16% +/- 1%. Standard Proctor ASTM...
-
Write an essay why your doctor should care about social Justice ( In the United States) Healthcare should be the focus with concrete example Please use the following sources in the essay: 1). Artiga,...
-
Use the following references to formulate your answer to the question below: American Hospital Association. (2012). Managing population health: The role of the hospital . Retrieved from...
-
Using Digital Tools to Enhance Your Job Search Just as electronic media have changed the way candidates seek jobs, these same digital tools are transforming the way employers select qualified...
-
Gourmet Juices is a company that sells a variety of natural fruit juices. The company has enjoyed record-breaking profits; its products have earned several local and international awards. However,...
-
Determine fundamental approaches to scientific research in addressing questions related to the natural world Develop questions about fundamental aspects of the natural world that inform personal...
-
Patrick is an Australian resident. What is LAMITO when taxable income equals $929772 for the year ending 30th June 2020
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
Benzene and hexane are being considered as solvents to extract acetic acid from aqueous mixtures. At 30C, distribution coefficients for the two solvents are K B = 0.098 mass fraction acetic acid in...
-
Draw and label the given streams and derive expressions for the indicated quantities in terms of labeled variables. The solution of Part (a) is given as an illustration. (a) A continuous stream...
-
Liquid water at 60 bar and 250C passes through an adiabatic expansion valve, emerging at a pressure Pf and temperature Tf. If Pf is low enough, some of the liquid evaporates. (a) If Pf = 10 bar,...
-
In the discussion at the beginning of this chapter, do you think the motivations for holding cash are reasonable?
-
Solicitous Bank offers your firm an 8 percent discount interest loan for up to \($3\) million, and in addition requires you to maintain a 6 percent compensating balance against the amount borrowed....
-
Formulate complete PDE problems (specify the equation, space domain, time interval, and boundary and initial conditions) for the following model situations. a) Conduction heat transfer occurs in a...

Study smarter with the SolutionInn App


