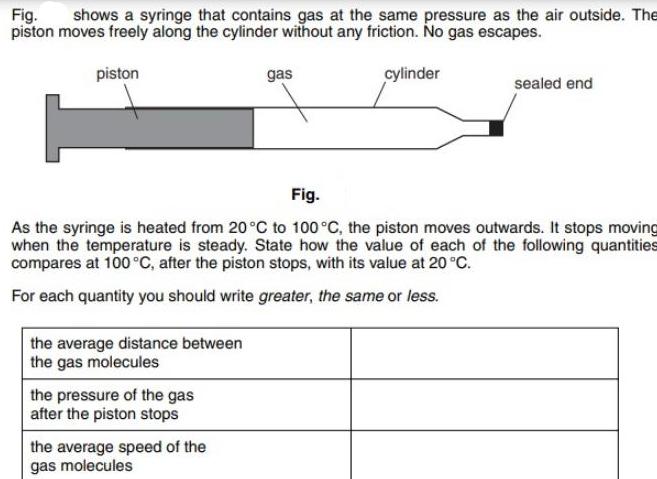
Fig. shows a syringe that contains gas at the same pressure as the air outside. The...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Fig. shows a syringe that contains gas at the same pressure as the air outside. The piston moves freely along the cylinder without any friction. No gas escapes. piston the average distance between the gas molecules the pressure of the gas after the piston stops gas Fig. As the syringe is heated from 20°C to 100 °C, the piston moves outwards. It stops moving when the temperature is steady. State how the value of each of the following quantities compares at 100 °C, after the piston stops, with its value at 20 °C. For each quantity you should write greater, the same or less. the average speed of the gas molecules cylinder sealed end Fig. shows a syringe that contains gas at the same pressure as the air outside. The piston moves freely along the cylinder without any friction. No gas escapes. piston the average distance between the gas molecules the pressure of the gas after the piston stops gas Fig. As the syringe is heated from 20°C to 100 °C, the piston moves outwards. It stops moving when the temperature is steady. State how the value of each of the following quantities compares at 100 °C, after the piston stops, with its value at 20 °C. For each quantity you should write greater, the same or less. the average speed of the gas molecules cylinder sealed end
Expert Answer:
Answer rating: 100% (QA)
This question is related to the behavior of gases in response to temperature changes based on the pr... View the full answer

Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Posted Date:
Students also viewed these physics questions
-
Problem 10: Provide your thoughts and examples on the following. As a guide a couple of paragraphs or about one side on one-page length for each one. a. Why is liquidity risk important? What factors...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Air is heated from 20 C to 800 C at constant pressure of 200 kPa in a cylinder with an initial volume of 4000 cm3. The entropy change, assuming an ideal gas with constant specific heats, is nearest:...
-
An important part of the customer service responsibilities of a cable company relates to the speed with which trouble in service can be repaired. Historically, the data show that the likelihood is...
-
Orr Electronics purchased a manufacturing plant four years ago for $4,500,000. The plant costs $1,000,000 per year to operate. Its current book value using straight-line depreciation is $3,500,000....
-
Fully explain why reduced values of R 1 in Fig. 17.11 will result in an increased angle of conduction. Fig. 17.11 R1. R A R1 0 90 90 conduction K (a) (b)
-
Find a form for a writ of attachment in California by viewing www.courts.ca.gov/forms.htm.
-
Wyco Park, a public camping ground near the Four Corners National Recreation Area, has compiled the following financial information as of December 31, 2019. Instructions (a) Determine Wyco Park's net...
-
Garcia Company can invest in one of two alternative projects. Project Y requires a $460,000 initial investment for new machinery with a four-year life and no salvage value. Project Z requires a...
-
Refer to Fig. 4.40. 8,0 in Semicircular hatch 10 in 26 in 20-in radius Ethylene glycol sg = 1.10 10 in Tank is symmetrical 30
-
Write the full (all electrons) electron configuration for an atom of gallium (Ga). Then state the number of valance electrons the atom contains, and in which energy level (n value) the valence...
-
Jing's BMX Incorporated has two service departments and three producing departments. Data for 2018 are as follows: Service Departments Maintenance Storeroom Producing Departments Machinery Assembly...
-
The initial cost of this investment is $550,000 and the operating and maintenance cost is $550,000 in year 1 increasing by $57,000 each year through year 12. Using 11% MARR, determine the equivalent...
-
2. The door to a castle is 3 m high by 2 m wide, with a mass of 100 kg. It has an iron door knob with mass 10 kg, 1.75 m from the hinge. The door is closed, but you pull on the doorknob with 100N of...
-
We know that profit is = TR - TC Total income is 18Q - 3q Total cost is 12+ Q What is the profit drop? What are the break even points of profit and cost When is the profit the highest and what is the...
-
Mika Corporation uses FIFO method to calculate the value of Inventory on hand on March 31 and cost of goods sold during March. Mar 1Beginning Inventory68 units @ P15.00 per unit 5Purchase140 units @...
-
You meet up with your friends, who started their used bookstore a year ago. At the time you gave them some advice. They have still not taken any management training. They are happy in their space...
-
Solve each equation or inequality. |6x8-4 = 0
-
One question that arises in phase equilibrium calculations and experiments is how many phases can be in equilibrium simultaneously, since this determines how many phases one should search for....
-
A considerable amount of methane is produced in Norway from its North Sea oil and gas wells. Some of this methane is converted to methanol by partial oxidation, and then biochemically converted to...
-
A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in the tank falls to 300 K. Assuming there is no heat transfer between the gas and the tank, find the pressure in...
-
What is the effect of pressure on equilibrium conversion of a gas-phase chemical reaction?
-
The diathermal wall (a) Is incapable of exchanging heat with the surroundings (b) Permits the full flow of heat from the system to the surroundings and vice versa (c) Both (a) and (b) (d) None of...
-
The total energy of a system comprises (a) Kinetic energy, potential energy and vibrational energy (b) Kinetic energy, potential energy and rotational energy (c) Kinetic energy, potential energy and...

Study smarter with the SolutionInn App


