Goal Solve for the efficiency of a heat engine using a five-step process the includes: 1....
Fantastic news! We've Found the answer you've been seeking!
Question:
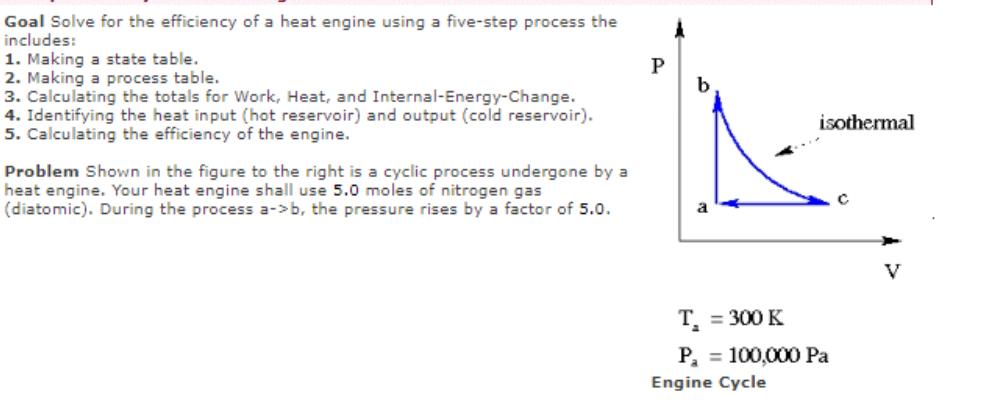

Transcribed Image Text:
Goal Solve for the efficiency of a heat engine using a five-step process the includes: 1. Making a state table. 2. Making a process table. 3. Calculating the totals for Work, Heat, and Internal-Energy-Change. 4. Identifying the heat input (hot reservoir) and output (cold reservoir). 5. Calculating the efficiency of the engine. Problem Shown in the figure to the right is a cyclic process undergone by a heat engine. Your heat engine shall use 5.0 moles of nitrogen gas (diatomic). During the process a->b, the pressure rises by a factor of 5.0. P A b isothermal T₁ = 300 K P₁ = 100,000 Pa Engine Cycle C V (1) Fill in the State Table (all pressures in Pascals, all volumes in cubic meters, all temperatures in K). (2) Fill in the Process Table (all entries in Joules). (3) Find the Totals: a b (5) Find the efficiency of the engine: U Pressure a-> b b->c c->a Work Work = Heat = dU = (4) Find the heat input (from "hot reservoir") and the Q-hot = heat output (to "cold reservoir"): Q-cold = efficiency Volume Heat J J J % Temperature du Goal Solve for the efficiency of a heat engine using a five-step process the includes: 1. Making a state table. 2. Making a process table. 3. Calculating the totals for Work, Heat, and Internal-Energy-Change. 4. Identifying the heat input (hot reservoir) and output (cold reservoir). 5. Calculating the efficiency of the engine. Problem Shown in the figure to the right is a cyclic process undergone by a heat engine. Your heat engine shall use 5.0 moles of nitrogen gas (diatomic). During the process a->b, the pressure rises by a factor of 5.0. P A b isothermal T₁ = 300 K P₁ = 100,000 Pa Engine Cycle C V (1) Fill in the State Table (all pressures in Pascals, all volumes in cubic meters, all temperatures in K). (2) Fill in the Process Table (all entries in Joules). (3) Find the Totals: a b (5) Find the efficiency of the engine: U Pressure a-> b b->c c->a Work Work = Heat = dU = (4) Find the heat input (from "hot reservoir") and the Q-hot = heat output (to "cold reservoir"): Q-cold = efficiency Volume Heat J J J % Temperature du
Expert Answer:
Answer rating: 100% (QA)
Step 1 Given that The number of moles gas is n3 At state A the pressure is P A 100000 P a the temper... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
A firebox is at 750 K, and the ambient temperature is 300 K. The efficiency of a Carnot engine doing 150 J of work as it transports energy between these constant-temperature baths is 60.0%. The...
-
The efficiency of a heat engine is to be improved by lowering the temperature of its low-temperature reservoir to a valuer l, below the environmental temperature l, by means of a refrigerator. The...
-
Review Organicos financial statements presented in the chapter for the month ended March 31, 2017. Review the balance sheet and determine the business form Hailey Walker has chosen to organize her...
-
Consider a failing bank. How much is a deposit of $350,000 worth if the FDIC uses the payoff method? The purchase and assumption method? Which method is more costly to taxpayers?
-
Consider the following facts to quantify the tax costs of various taxable acquisition structures when the target is a freestanding C corporation. Wolverine, Inc., wants to purchase Reel Deal, Inc.,...
-
Give some examples of requirements where they define both objectives and constraints.
-
Doritos used a guerrilla marketing campaign in which product images were projected on the sides of buildings while inviting viewers to text message their flavor preference. How do such tactics fit...
-
Translate the following code into MIPS code. int Sum (int a) { } if (a <3) return (1); else return (a + Sum (a - 2) + Sum (a-3));
-
Write a query to display the patron ID, book number, and days kept for each checkout. Days Kept is the difference from the date on which the book is returned to the date it was checked out. Sort the...
-
recursively defined by Create a program for a scientific calculator for finding the terms of the sequence (pn) Pn-1-Pn-2 Pn=Pn-1-f(Pn-1). f(Pn-1)-f(pn-2) for all n 2, where and where the function f...
-
Assume your organization has the following inventory changes during the year: Beginning Inventory January purchases April purchases Total Units Used 20 units valued at $\$ 7,500$ each 10 units at $\$...
-
Why would you want to keep inventory levels low?
-
How does inflation impact the valuation of an organization's inventory?
-
What are some uses of budgets for control?
-
What are some issues that must be considered regarding payroll policy?
-
1. in 2000, the nominal gdp growth of a country was 8% and the real gdp growth was 4%. what was the rate of inflation for this country? 0 2 08
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
Derive (39), which is the form of the law of mass action when n is no longer small compared to nc.
-
Consider a system at temperature , with N atoms of mass M in volume. Let (0) denote the value of the chemical potential at the surface of the earth (a) Prove carefully and honestly that the value of...
-
It has been said* that set to strum unintelligently on the typewrites for millions of years, would be bound in time to write all the books in the British Museum. This statement is nonsense, for it...
-
Define and distinguish between an open and a closed adoption.
-
Define and distinguish between an independent and an agency adoption.
-
Define and give an example of an equitable adoption.

Study smarter with the SolutionInn App


