DIRECTIONS: 1. There are two parts to this group activity: a) Symbol and Calculated Atomic Mass...
Fantastic news! We've Found the answer you've been seeking!
Question:
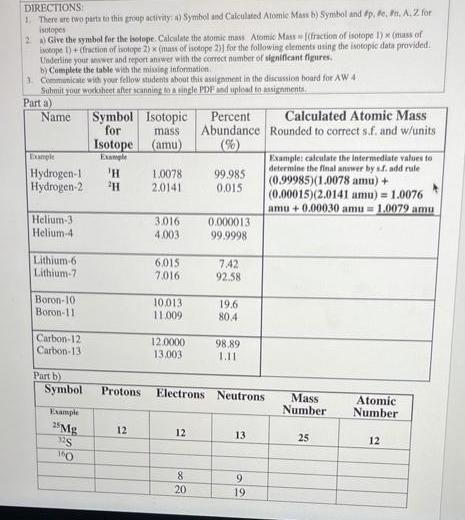
Transcribed Image Text:
DIRECTIONS: 1. There are two parts to this group activity: a) Symbol and Calculated Atomic Mass b) Symbol and #p, #e, #n, A. 2 for isotopes 2) Give the symbol for the isotope. Calculate the atomic mass Atomic Mass [(fraction of isotope 1) x (mass of isotope 1)+ (fraction of isotope 2) x (mass of isotope 2)] for the following elements using the isotopic data provided. Underline your answer and report answer with the correct number of significant figures. b) Complete the table with the missing information. 3. Communicate with your fellow students about this assignment in the discussion board for AW 4 Submit your worksheet after scanning to a single PDF and upload to assignments. Parta) Name Helium-3 Helium-4 Example Hydrogen-1 'H Hydrogen-2 ¹H Lithium-6. Lithium-7 Boron-10: Boron-11 Carbon-12 Carbon-13 Part b) Symbol Symbol for Example 25 Mg 32S 160 Isotope Example Protons 12 Isotopic mass. (amu) 1.0078 2.0141 3.016 4.003 6.015 7.016 10.013. 11.009 12.0000 13,003 12 Percent Abundance (%) 8 20 99.985 0.015 0.000013 99.9998 7.42 92.58 19.6 80.4 Electrons Neutrons 98,89 1.11 13 9 56 19 Calculated Atomic Mass Rounded to correct s.f. and w/units Example: calculate the intermediate values to determine the final answer by sf. add rule (0.99985)(1.0078 amu) + (0.00015)(2.0141 amu) = 1.0076 amu+0.00030 amu= 1.0079 amu Mass Number 25 Atomic Number 12 DIRECTIONS: 1. There are two parts to this group activity: a) Symbol and Calculated Atomic Mass b) Symbol and #p, #e, #n, A. 2 for isotopes 2) Give the symbol for the isotope. Calculate the atomic mass Atomic Mass [(fraction of isotope 1) x (mass of isotope 1)+ (fraction of isotope 2) x (mass of isotope 2)] for the following elements using the isotopic data provided. Underline your answer and report answer with the correct number of significant figures. b) Complete the table with the missing information. 3. Communicate with your fellow students about this assignment in the discussion board for AW 4 Submit your worksheet after scanning to a single PDF and upload to assignments. Parta) Name Helium-3 Helium-4 Example Hydrogen-1 'H Hydrogen-2 ¹H Lithium-6. Lithium-7 Boron-10: Boron-11 Carbon-12 Carbon-13 Part b) Symbol Symbol for Example 25 Mg 32S 160 Isotope Example Protons 12 Isotopic mass. (amu) 1.0078 2.0141 3.016 4.003 6.015 7.016 10.013. 11.009 12.0000 13,003 12 Percent Abundance (%) 8 20 99.985 0.015 0.000013 99.9998 7.42 92.58 19.6 80.4 Electrons Neutrons 98,89 1.11 13 9 56 19 Calculated Atomic Mass Rounded to correct s.f. and w/units Example: calculate the intermediate values to determine the final answer by sf. add rule (0.99985)(1.0078 amu) + (0.00015)(2.0141 amu) = 1.0076 amu+0.00030 amu= 1.0079 amu Mass Number 25 Atomic Number 12
Expert Answer:
Answer rating: 100% (QA)
Partla Name Symbol Isotopic Percent mass amu Abundance 1 H Hydrogin1 Hydrogen2 2H ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
please help explain and answer study question number 2 & 3 page discussion questions 1. refer to the fundamental principles governing an audit (see chapter 2@ ). under the responsibili
-
Discuss the meaning of this mathematical expression and verify it using EXCEL = (1 + r)4 FV4 PVo = FV4 i.e. a) Explain what is the meaning of the ratio PVO b) Explain what is the meaning of the term...
-
a. Find the nth-order Taylor polynomials for the given function centered at the given point a, for n = 0, 1, and 2.b. Graph the Taylor polynomials and the function. f(x) = ln x, a e =
-
Net Suite Inc. is a leading provider of on-demand enterprise applications, including CRM as featured in the case. Other important players in this market include Salesforce.com and Siebel on Demand....
-
1. What are the issues in this case? In what order should Andy Chin and Adeline Koh address them? 2. Should Koh approve the packaging machine investment? How did you analyze this issue? In July 2001,...
-
What the differences are between federal and state court jurisdiction?
-
The total wages and salaries earned by all employees of Langen Electronics, Ltd. during March, as shown in the labor cost summary and the schedule of fixed administrative and sales salaries, are...
-
Sarasota Industrial Inc. of Florida purchased computer equipment from Sydney Tech. of Australia for A$ 5 , 0 0 0 , 0 0 0 with payment due in 3 months. The forecasting department of the firm expects...
-
Question A: Stay Safe International manufactures industrial safety equipment at its plant in Evans- ville, Indiana. The company has initiated DRP to coordinate finished goods distribution from the...
-
A manufacturer uses normal costing and allocates manufacturing overhead costs to products on the basis of Machine hours in the Machining Department and on the basis of Direct labor hours in the...
-
Drug 4 mg I every 3 hours prn pain. Supplied as 10mg/10 ml. Calculate mL per dose?
-
Chrome File Edit View History Bookmarks Profiles Tab Window Help MT217 Finance O PG Campus =Quiz: Unit 6 Lab Quiz X PG Campus G Sir W. Arthur Lewi... *FREE Digital Down... Christopher William X $ Do...
-
How to Build a demand table utilizing a generalized function. The demand generalization function for a product is as follows: Qd=200-10p-8M-10PR 1. Simplify the function when M-100 and PR=200 2....
-
For the previous 3 years, the board of directors has declared no dividends. This year they declared a $240,000 dividend. There are 5,000 shares of common stock and 8,000 shares of 7%, $100 par value...
-
How can one tell many months the positive skew had in it?
-
Jim Foster is the owner of Selwyn Pub in North Carolina. Jim's family was the owner of the pub for over four decades. The distinguishing feature of the Selwyn Pub is the massive oak tree in front of...
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
Define cost tracing, cost allocation, allocation base and cost driver.
-
PTP produces two products from different combinations of the same resources. Details of the selling price and costs per unit for each product are shown below: The fixed costs of the company are $50...
-
N Ltd?s chief executive believes the company is holding excessive stocks and has asked for the management accountant to carry out an investigation.Information on the two stock items is given below:...
-
Which one of the following is an exact straight line mechanism using lower pairs? (a) Watt's mechanism (b) Grasshopper mechanism (c) Robert's mechanism (d) Peaucellier's mechanism.
-
In the epicyclic gear train shown in the Fig. \(88, T_{A}=40, T_{B}=20\). For three revolutions of the arm, the gear \(B\) will rotate through (a) 6 revolutions (b) 2.5 revolutions (c) 3 revolutions...
-
Which one of the following statements in respect of involute profiles for gear teeth is not correct? (a) Interference occurs in involute profiles (b) Involute tooth form is sensitive to change in...

Study smarter with the SolutionInn App


