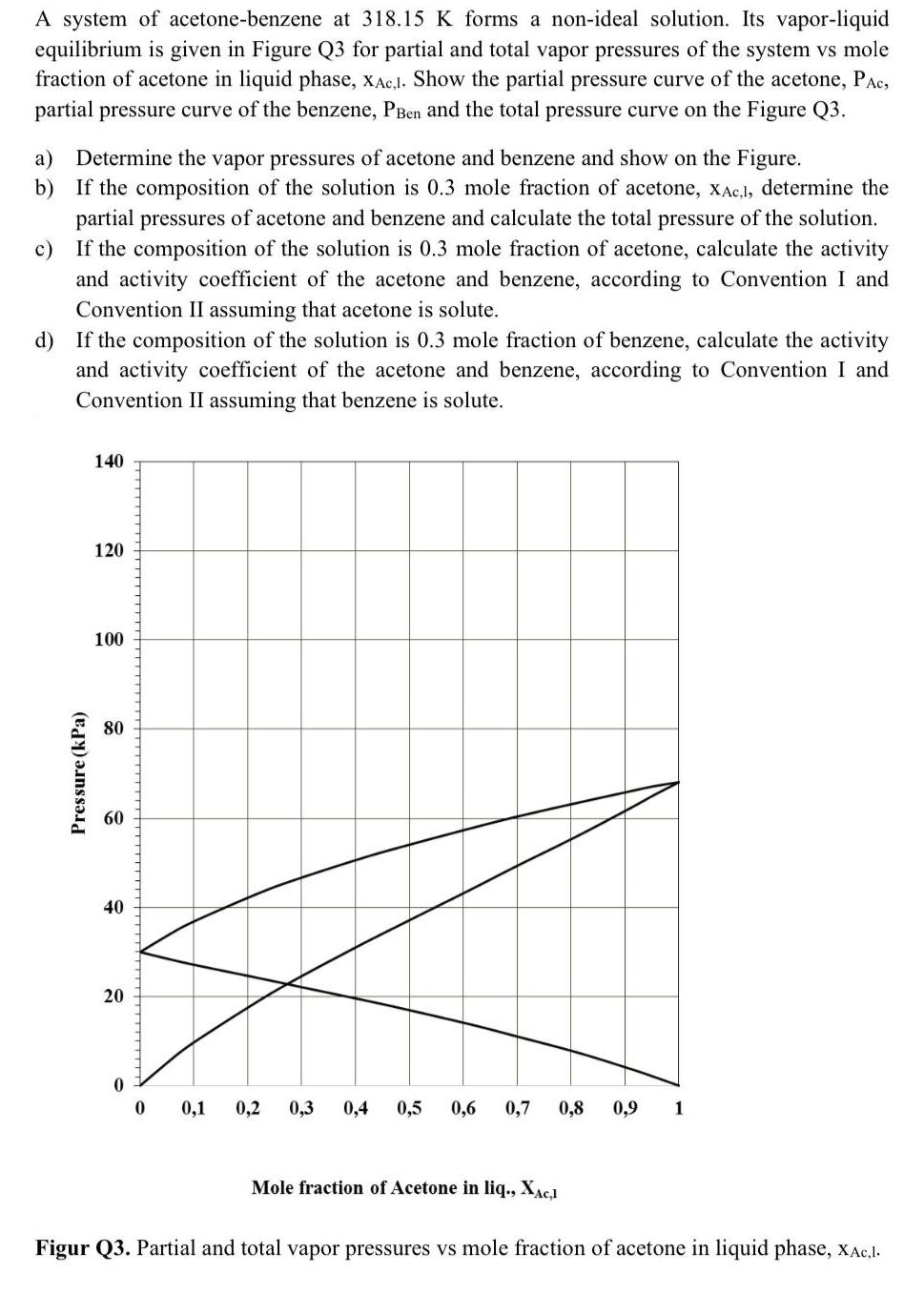
A system of acetone-benzene at 318.15 K forms a non-ideal solution. Its vapor-liquid equilibrium is given...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A system of acetone-benzene at 318.15 K forms a non-ideal solution. Its vapor-liquid equilibrium is given in Figure Q3 for partial and total vapor pressures of the system vs mole fraction of acetone in liquid phase, xAc,I. Show the partial pressure curve of the acetone, PAc, partial pressure curve of the benzene, PBen and the total pressure curve on the Figure Q3. a) Determine the vapor pressures of acetone and benzene and show on the Figure. b) If the composition of the solution is 0.3 mole fraction of acetone, xAc,l, determine the partial pressures of acetone and benzene and calculate the total pressure of the solution. If the composition of the solution is 0.3 mole fraction of acetone, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and c) Convention II assuming that acetone is solute. d) If the composition of the solution is 0.3 mole fraction of benzene, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and Convention II assuming that benzene is solute. 140 120 100 80 60 40 20 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Acetone in liq., XAC1 Figur Q3. Partial and total vapor pressures vs mole fraction of acetone in liquid phase, xAc,1. Pressure (kPa) A system of acetone-benzene at 318.15 K forms a non-ideal solution. Its vapor-liquid equilibrium is given in Figure Q3 for partial and total vapor pressures of the system vs mole fraction of acetone in liquid phase, xAc,I. Show the partial pressure curve of the acetone, PAc, partial pressure curve of the benzene, PBen and the total pressure curve on the Figure Q3. a) Determine the vapor pressures of acetone and benzene and show on the Figure. b) If the composition of the solution is 0.3 mole fraction of acetone, xAc,l, determine the partial pressures of acetone and benzene and calculate the total pressure of the solution. If the composition of the solution is 0.3 mole fraction of acetone, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and c) Convention II assuming that acetone is solute. d) If the composition of the solution is 0.3 mole fraction of benzene, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and Convention II assuming that benzene is solute. 140 120 100 80 60 40 20 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Acetone in liq., XAC1 Figur Q3. Partial and total vapor pressures vs mole fraction of acetone in liquid phase, xAc,1. Pressure (kPa)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The current in a 50-mH inductor is given in Figure P5.29. Sketch the inductor voltage. 100 :/68 10 t(ms) -100 1-- Figure P5.29
-
A Nichols chart is given in Figure E9.14 for a system with Gc(jÏ)G(jÏ)). Using the following table, find (a) the peak resonance MpÏ) in dB; (b) the resonant frequency cor; (c) the 3-dB...
-
An enthalpy-concentration diagram is given in Figure for a mixture of n-hcxanc (H). and n-octane (0) at 101 kPa. Using this diagram, determine the following: (a) The mole-fraction composition of the...
-
If you were in charge of B&D, indicate the possible decisions you might take as regards the four Ps and the points you would want to keep in mind as you go about the task of marketing for...
-
On January 4, 2015, an FI has the following balance sheet (rates = 10 percent) DGAP = [6 (170/200)4] = 2.6 years > 0 The FI manager thinks rates will increase by 0.75 percent in the next three...
-
How could opinion shopping be (a) Suspect or (b) Helpful?
-
Diageo North America, Inc., the owner of the Bulleit brand family of whiskeys, sued competitor W.J. Deutsch & Sons Ltd. for allegedly infringing upon the trade dress of Diageos Bulleit bottles....
-
Hillyard Company, an office supplies specialty store, prepares its master budget on a quarterly basis. The following data have been assembled to assist in preparing the master budget for the first...
-
Identify the entities and the relationships the diagram given below Translate the Diagram Into Relational Model Fname Minit Lname San Bdate Sex No Street Apt no City State Zip Name PERSON Address...
-
A researcher at a marketing firm examines whether the age of a consumer matters when buying athletic clothing. Her initial feeling is that Brand A attracts a younger customer, whereas the more...
-
You work for Bryan Medical Billing & Coding Inc. Early Monday morning, your executives call you into a meeting to go over some key initiatives they hope to achieve this quarter. You're tasked with...
-
Vandelay Industries has a target capital structure consisting of 30% debt, 10% preferred stock, and 60% common equity. Vandelay has 20-year, 12% semiannual coupon bonds that sell at their par value...
-
A perpetuity is having steady cash flow. Year 1 payment is $1,000 and it will continue to go for forever on a yearly basis. What is the present value of this perpetuity at 10% rate.
-
Over the last six years the shares of company XYZ's stock had returns of 18 percent, 19 percent, 10 percent, 8 percent, 18 percent, and -7 percent. Required: (a)Calculate the arithmetic average...
-
A microphone factory wants to schedule order lot sizes for microphone grid. The anticipated demand for this item over the next 12 months start from Jan 2022 until Dec 2022 is 81.300, 23.040, 12.396,...
-
Dr. Gregory is calculating his operating margin to determine how his cardiology practice compares to competitors in the area. Dr. Gregory's total revenue is $975,000 and the cardiology practice's net...
-
Kamal is a lead independent director in a company. The role played by him is/are: a. To improve corporate governance standards of the company by working as a watchdog. b. To direct the company with...
-
Michelles trust is subject to 3.8% surtax on the lesser of the trusts net investment income or the excess of the trusts adjusted gross income over the $12,400 threshold (the highest trust tax rate)....
-
Consider a unity negative feedback system with where K is set equal to 100 in order to achieve a specified Kv = 2. We wish to add a lead-lag compensator Show that the gain margin of the compensated...
-
Consider the system in state variable form x = Ax + Bu y = Cx + Du with (a) Compute the transfer function G(s) = Y(s)/U(s). (b) Determine the poles and zeros of the system. (c) If possible, represent...
-
The dynamics of a rocket are represented by and state variable feedback is used, where = - x1 - 25x2 + r.. Determine the roots of the characteristic equation of this system and the response of the...
-
An engineering organisational system is composed of major groups such as management, research and development, preliminary design, experiments, product design and drafting, fabrication and...
-
The student-teacher learning activity is inherently a feedback exercise intended to reduce the system error to a minimum. The desired output is the knowledge being studied, and the student is the...
-
Give two examples of feedback control systems in which a human acts as a controller.

Study smarter with the SolutionInn App


