Match the following IR spectra to the corresponding molecule: %T 100 80 60 40 20 9000...
Fantastic news! We've Found the answer you've been seeking!
Question:
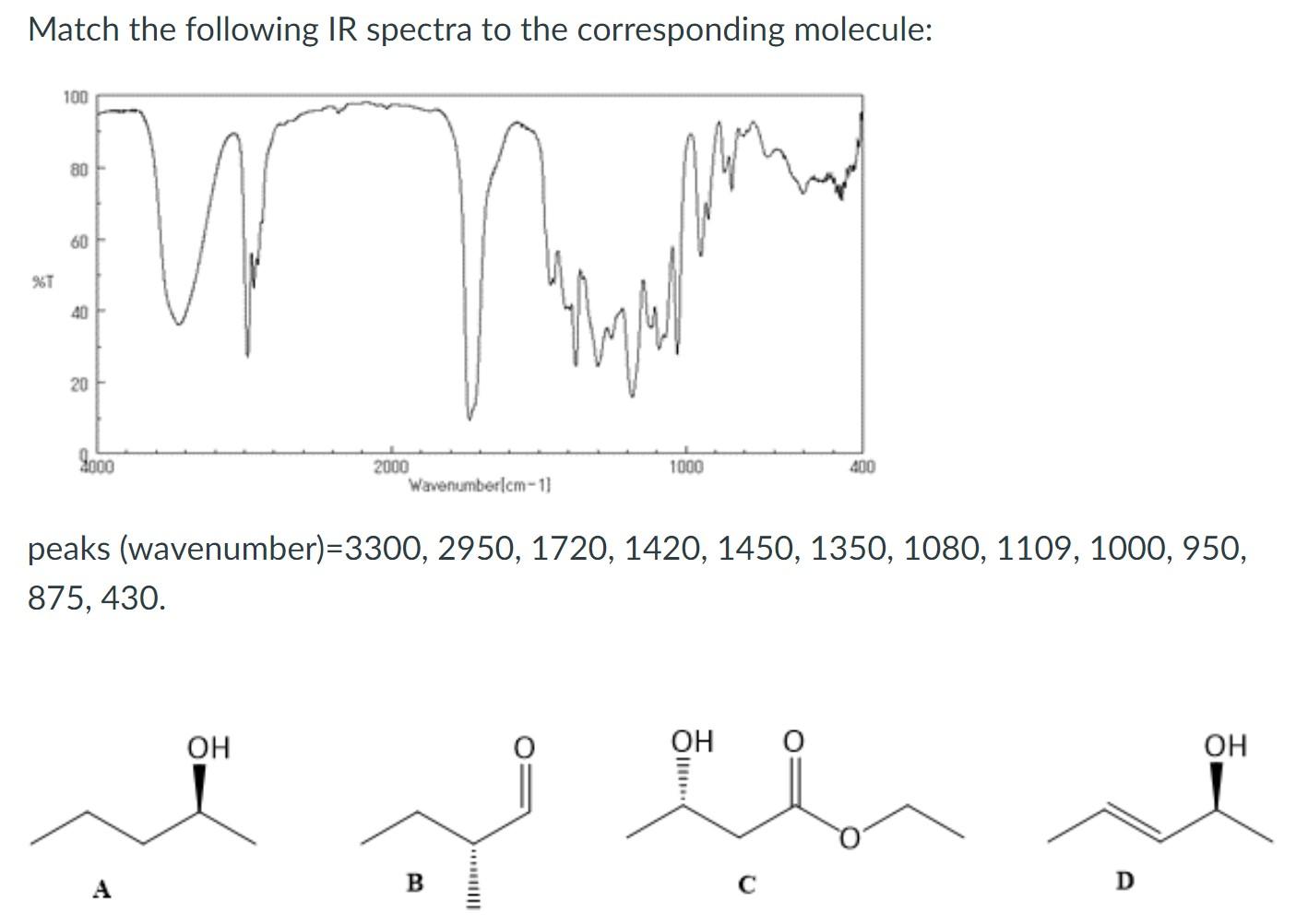
Transcribed Image Text:
Match the following IR spectra to the corresponding molecule: %T 100 80 60 40 20 9000 Butt OH 2000 Wavenumberlcm-1] 1000 peaks (wavenumber)=3300, 2950, 1720, 1420, 1450, 1350, 1080, 1109, 1000, 950, 875, 430. B OH 400 C D OH Match the following IR spectra to the corresponding molecule: %T 100 80 60 40 20 9000 Butt OH 2000 Wavenumberlcm-1] 1000 peaks (wavenumber)=3300, 2950, 1720, 1420, 1450, 1350, 1080, 1109, 1000, 950, 875, 430. B OH 400 C D OH
Expert Answer:
Answer rating: 100% (QA)
Sol Functional group 0H Stretching Sp C stretch Ester stretching C0R Alkane CHbendi... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
4. Reactivity considerations suggest that an unknown compound, which has the mass spectrum shown in Fig. 2, is a vinyl halide. Combustion analysis gave the following results: Carbon, 38.89 %;...
-
Total Transaction Count 140 120 100 80 60 40 20 0 0 SUMMARY OUTPUT ANOVA Regression Statistics Multiple R R Square Adjusted R Square Standard Error Observations y = -0.004x + 69.283 R = 2E-06...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
Jane Doe is single and claims 2 withholding allowances. If her weekly gross earning is $1,450, then determine the Federal Withholding Tax.
-
A stock has an expected rate of return of 4%. What is its beta? Assume that the risk-free rate of interest is 6% and the expected rate of return on the market is 16%.
-
For a stirred-tank heater, assume the transfer function between the heater input change u(t) (cal/sec) and the tank temperature change y(t)( C) can be modeled as G(s) = 5 / 3s + 1 (a) Using the...
-
Crawford-Austin Properties included the following stockholders' equity on its year-end balance sheet at December 31, 2006. Requirements 1. Identify the different issues of stock that Crawford-Austin...
-
Lanza Research Inc. manufactures high-quality hair care products in California. Copyrighted labels are attached to all products and packaging. In the United States, Lanza sells exclusively to...
-
Manitoba Exporters Inc. (MEI) sells Inuit carvings to countries throughout the world. On December 1, Year 5, MEI sold 10,000 carvings to a wholesaler in a foreign country at a selling price of...
-
On October 1, 20Y8, Jacinto Suarez and Tricia Fritz form a partnership. Suarez agrees to invest $25,000 in cash and inventory valued at $60,000. Fritz invests certain business assets at valuations...
-
Draw and annotate the shear force and bending moment diagrams for the frame. Show calculations for the shear force and bending moment values at critical points along the frame. Selena works at...
-
You are an insurance investigator called out to investigate a claim based on a patient slipping and falling inside of a hospital. What are the initial steps as part of your investigation? What could...
-
Why does Law of Demand and Supply contribute to profit-maximizing management decision-making?
-
When referring to social welfare and organizational context in generalist practice, what are we referring to in social work?
-
What is the age rule for the Child Tax Credit for Tax Year 2022?
-
The new business models appearing in today's environment call for organizations to have a solid understanding of: A . Cost and utilization of healthcare resources B . Revenue optimization and cost C...
-
Prepare a CRITICAL ANALYSIS PAPER on the essay "The Political Economy of Gross Domestic Product (GDP) Accounting and the Philippine Case" written by Prof. Edilberto M. Villegas. Please see file...
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure 13-41 (page 603) to estimate the chemical shifts. (a) (b) (c) (d) C CH H,C CH C-H C-C
-
(a) Explain how pyrrole is isoelectronic with the cyclopentadienyl anion. (b) Specifically, what is the difference between the cyclopentadienyl anion and pyrrole? (c) Draw resonance forms to show the...
-
Predict the products of saponification of the following esters. (a) (b) (c) (d) CH CHC-OCH-CH
-
A stirrer-container assembly contains a certain amount of fluid. The stirrer performs \(3 \mathrm{hp}\) work on the system. The heat developed by stirring is \(4000 \mathrm{~kJ} / \mathrm{h}\) and is...
-
Derive the mathematical expression of the first law of thermodynamics.
-
A system consisting of a gas confined in a cylinder undergoes a series of processes shown in Fig. 2.11. During the process A-1-B, \(70 \mathrm{~kJ}\) of heat is added while it does 45 \(\mathrm{kJ}\)...

Study smarter with the SolutionInn App


