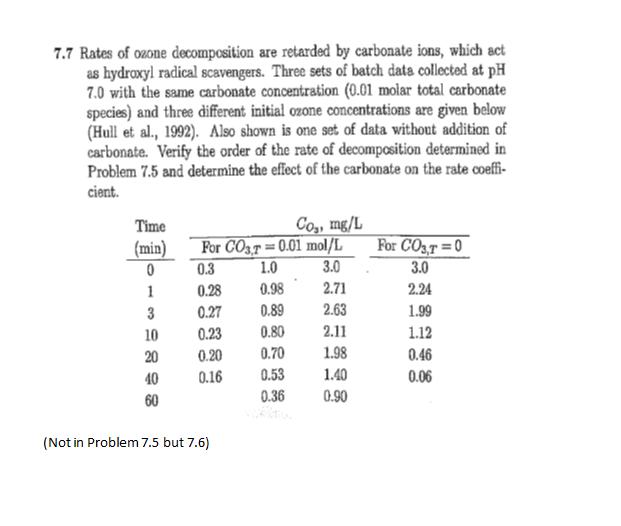
7.7 Rates of ozone decomposition are retarded by carbonate ions, which act as hydroxyl radical scavengers....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
7.7 Rates of ozone decomposition are retarded by carbonate ions, which act as hydroxyl radical scavengers. Three sets of batch data collected at pH 7.0 with the same carbonate concentration (0.01 molar total carbonate species) and three different initial ozone concentrations are given below (Hull et al., 1992). Also shown is one set of data without addition of carbonate. Verify the order of the rate of decomposition determined in Problem 7.5 and determine the effect of the carbonate on the rate coeffi- cient. Time (min) 0 1 3 10 20 40 60 For CO3,7= 0.01 mol/L 0.3 1.0 3.0 0.98 2.71 0.89 2.63 0.80 2.11 0.70 1.98 0.53 1.40 0.36 0.90 0.28 0.27 0.23 0.20 0.16 Co₂, mg/L (Not in Problem 7.5 but 7.6) . For CO3,T=0 3.0 2.24 1.99 1.12 0.46 0.06 7.7 Rates of ozone decomposition are retarded by carbonate ions, which act as hydroxyl radical scavengers. Three sets of batch data collected at pH 7.0 with the same carbonate concentration (0.01 molar total carbonate species) and three different initial ozone concentrations are given below (Hull et al., 1992). Also shown is one set of data without addition of carbonate. Verify the order of the rate of decomposition determined in Problem 7.5 and determine the effect of the carbonate on the rate coeffi- cient. Time (min) 0 1 3 10 20 40 60 For CO3,7= 0.01 mol/L 0.3 1.0 3.0 0.98 2.71 0.89 2.63 0.80 2.11 0.70 1.98 0.53 1.40 0.36 0.90 0.28 0.27 0.23 0.20 0.16 Co₂, mg/L (Not in Problem 7.5 but 7.6) . For CO3,T=0 3.0 2.24 1.99 1.12 0.46 0.06
Expert Answer:
Answer rating: 100% (QA)
Solution Plotting each group of rate data as 1reaction time versus In03 wil... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Set out below are ratios relating to three different businesses. Each business operates within a different industrial sector. Required: State, with reasons, which of the above is: (a) A holiday tour...
-
Below are shown three different crystallographic planes for a unit cell of some hypothetical metal. The circles represent atoms: (a) To what crystal system does the unit cell belong? (b) What would...
-
Below you will find three data sets that have already been sorted. Fill in the blanks to find the upper and lower quartiles. Measurements Above and Below and Measurements Above and Below and Position...
-
What is the difference between average product and marginal product? Can you sketch a total product function such that the average and marginal product functions coincide with each other?
-
Paul Smith is married and filing his tax jointly with his wife, Anna. Their combined taxable income is $167,983. Use the tax computation worksheet (Table 20-2) to calculate the tax they must pay?
-
A Lenoir engine (described in P3.5) operates with inlet conditions of \(p_{1}=1 \mathrm{bar}\) and \(T_{1}=27^{\circ} \mathrm{C}\). The energy added to the charge is \(1000 \mathrm{~kJ} /...
-
Why do welfare programs create work disincentives?
-
The following issues are related to the accuracy and reliability of financial plans. Explain the process/issues related to each. Top-down versus bottom-up planning Plans as statements of goals...
-
The department head has the impression that some employees are less satisfied with their manager if the employee has worked in other companies. What is the correlation between Number of Companies...
-
For each of the following scenarios, indicate which of the four basic tax planning variables (entity, character, time period, jurisdiction) impacts after-tax value. Note that more than one variable...
-
Suppose that =1 = -3 and y = -2 when x = 1. What is the approximate value of when x = 0.99? () 2.03 (b) 1.97 () 5 (d) 0.03 (e) none of these
-
a wholly owned subsidiary of Trumaine Corporation, generated a $ 80 0 , 0 0 0 taxable loss in its first year of operations. True s activities and sales are restricted to State A , which imposes...
-
Zina Chaburi has a full time job as a nurse in her local hospital. In her spare time she has a goat farming operation. The goat farm began in 2 0 2 1 , which resulted in a loss of $ 1 0 , 0 0 0 . She...
-
At the beginning of Year 4 , Able Company had the following portfolio of investments in trading securities ( all of which were acquired at par value ) : Security Cost 1 / 1 / Y 4 Fair Value A $ 2 0 ,...
-
Lord Peter Wimsey, a Bermudian citizen and resident, has real property holdings in the United States. The holdings consist of a fee ownership interest in ten real estate parcels in Jacksonville,...
-
Cullumber Company is considering an investment that will return a lump sum of $ 5 8 0 , 0 0 0 5 years from now. What amount should Cullumber pay for this investment in order to earn a 4 % return?
-
discuss how business owners, managers, and other organizational leaders use the four basic financial statements to make business decisions. Your discussion should include: A description of the four...
-
Ask students to outline the reasons why the various elements of culture (social structures and control systems, language and aesthetics, religion and other belief systems, educational systems, etc.)...
-
The designations 1A through 8A used for certain families of the periodic table are helpful for predicting the charges on ions in binary ionic compounds. In these compounds, the metals generally take...
-
The standard enthalpy of formation of H2O(l) at 298 K is 285.8 kJ/ mol. Calculate the change in internal energy for the following process at 298 K and 1 atm: H2O(l) H2(g) + O2(g) Eo = ?
-
Consider the following galvanic cell: Calculate the Ksp value for Ag2SO4(s). Note that to obtain silver ions in the right compartment (the cathode compartment), excess solid Ag2SO4 was added and some...
-
Consider the situation illustrated in Figure 25. 11. A positively charged particle is lifted against the uniform electric field of a negatively charged plate. Ignoring any gravitational interactions,...
-
A positively charged particle is moved from point A to point B in the electric field of the massive, stationary, positively charged object in Figure 25. 12. (a) Is the electrostatic work done on the...
-
Figure 25. 13 shows both the electric field lines and the equipotentials associated with the given charge distribution. (a) Is the potential at point A higher than, lower than, or the same as the...

Study smarter with the SolutionInn App


