The analysis of a compound gives the following percent composition by mass: C: 61.49 percent; H:...
Fantastic news! We've Found the answer you've been seeking!
Question:
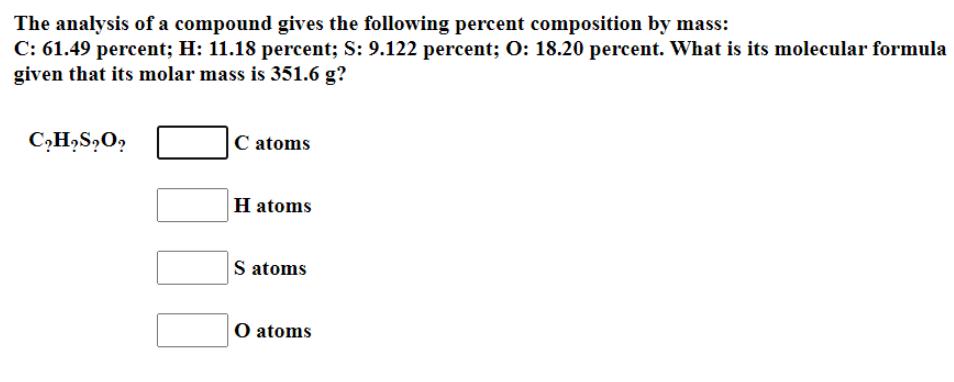
Transcribed Image Text:
The analysis of a compound gives the following percent composition by mass: C: 61.49 percent; H: 11.18 percent; S: 9.122 percent; O: 18.20 percent. What is its molecular formula given that its molar mass is 351.6 g? C₂H₂S₂O₂ C atoms toms H atoms S atoms O atoms The analysis of a compound gives the following percent composition by mass: C: 61.49 percent; H: 11.18 percent; S: 9.122 percent; O: 18.20 percent. What is its molecular formula given that its molar mass is 351.6 g? C₂H₂S₂O₂ C atoms toms H atoms S atoms O atoms
Expert Answer:
Answer rating: 100% (QA)
Bo to find Molecular formula given that its mase 3516 g molar Let there is w... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Allicin is the compound responsible for the characteristic smell of garlic. An analysis of the compound gives the following percent composition by mass: C: 44.4 percent; H: 6.21 percent; S: 39.5...
-
Peroxyacylnitrate (PAN) is one of the components of smog. It is a compound of C, H, N, and O. Determine the percent composition of oxygen and the empirical formula from the following percent...
-
The SEC criticized Frank Sinopoli for not sending an audit inquiry letter to Geo Securities external legal counsel. Describe the nature and purpose of such a letter. Do you agree with the SEC that...
-
Terrorists often use wireless networks to communicate. To disrupt these communications, the U.S. military uses jamming attacks on the wireless networks. The International Journal of Production...
-
Lets look at Target, one of the largest retailers found throughout the United States. Think about Target, all its stakeholders , what products it sells, and how and where it sells its products. Think...
-
What is a double-barreled question?
-
1. What effect does the outcome of this case have on Redmonds ability to earn a living? Should PepsiCo have to re-hire him? 2. Suppose Redmond had been terminated by PepsiCo before he was hired by...
-
Midnight Sun Apparel Company uses normal costing, and manufacturing overhead is applied to work-in-process on the basis of machine hours. On January 1 of the current year, there were no balances in...
-
Knockoffs Unlimited, a nationwide distributor of low-cost imitation designer necklaces, has an exclusive franchise on the distribution of the necklaces, and sales have grown so rapidly over the past...
-
ABC Company has a number of customers in the province who usually pay on time but because of the mailing time, processing time and clearing time, cash cannot be obtained as payment is made. A bank...
-
Explain the cohort effects in explaining differences in (LPPR) for men and women from this figure. Explain the differences in LFPR associated with belonging to a particular generation. Explain this...
-
A random variable x has the following probability distribution. | | f(x) 0 0.35 1 0.25 2 0.27 3 0.08 4 0.05 (a) Determine the expected value of x. (b) Determine the variance.
-
List and describe two ways (one for demand and one for supply) to address positive consumption externality. How would they be effective? What are the two possible "cons" of these 2 ways?
-
Working Capital and Short Term Liquidity Ratios Bell Company has a current ratio of 2.85 on December 31. On that date the company's current assets are as follows: Cash Short-term investments $16,400...
-
Provide an analysis of the state's possible position on the three federal policies or ideas regarding States that are mandated to provide tuition-free education at community colleges, with the...
-
The S&OP team at Kansas Fumiture, has received estimates of demand requirements as shown in the table. Assuming one-time stockout costs for lost sales of $100 per unit, inventory carrying costs of...
-
What services are provided by the provincial and territorial governments?
-
A mixture of Na2CO3 and MgCO3 of mass 7.63 g is reacted with an excess of hydrochloric acid. The CO2 gas generated occupies a volume of 1.67 L at 1.24 atm and 26C. From these data, calculate the...
-
In 2.00 min, 29.7 mL of He effuse through a small hole. Under the same conditions of pressure and temperature, 10.0 mL of a mixture of CO and CO2 effuse through the hole in the same amount of time....
-
Give the chemical names of the following organic compounds and write their formulas: marsh gas, grain alcohol, wood alcohol, rubbing alcohol, antifreeze, mothballs, chief ingredient of vinegar.
-
Figure18 . 25 shows the series of Walmart daily closing prices between February 2001 and February 2002 (thanks to Chris Albright for suggesting the use of these data, which are publicly available,...
-
Shipments of Household Appliances. The time plot in Figure 18 . 31 shows the series of quarterly shipments (in million dollars) of US household appliances between 1985 and 1989 (dataare available in...
-
Relation Between Moving Average and Exponential Smoothing. Assume that we apply a moving average to a series, using a very short window span. If we wanted to achieve an equivalent result using simple...

Study smarter with the SolutionInn App


