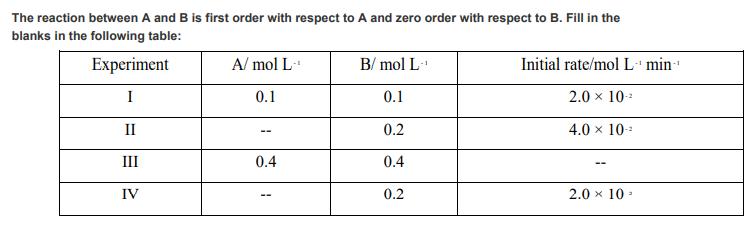
The reaction between A and B is first order with respect to A and zero order...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The reaction between A and B is first order with respect to A and zero order with respect to B. Fill in the blanks in the following table: Experiment I II III IV A/mol L- 0.1 0.4 1 B/mol L- 0.1 0.2 0.4 0.2 Initial rate/mol L min 2.0 10- 4.0 10- 2.0 10. The reaction between A and B is first order with respect to A and zero order with respect to B. Fill in the blanks in the following table: Experiment I II III IV A/mol L- 0.1 0.4 1 B/mol L- 0.1 0.2 0.4 0.2 Initial rate/mol L min 2.0 10- 4.0 10- 2.0 10.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Using the DET2TL program, recommend the most profitable configuration of a two-machine deterministic processing time line. The machines are the same. Now, there is no target production rate. Instead,...
-
Fill in the blanks in the following table of GDPstatistics: 2005 2006 2007 13,843 Nominal GDP 12,434 Real GDP 11,003 11,319 GDP deflator 116.6 119.7
-
A rate law is one-half order with respect to a reactant. What is the effect on the rate when the concentration of this reactant is doubled?
-
The slurry of Prob. 29.3 is to be filtered in a press having a total area of 8 m 2 and operated at a constant pressure drop of 2 atm. The frames are 36 mm thick. Assume that the filter medium...
-
Inventors require a 15% rate of return on brooks sisterss stock (rs = 15%). (a) What would the value of Brooks stock be if the previous dividend was D0 = $2 and if investors expect dividends to grow...
-
The chart shows approximations of the density of metals, d, in grams per cubic centimeters for six metals. The mass of an object can be found by using the formula m = d V, in which m is the mass of...
-
The Arcadia Company made an offer to purchase all of the outstanding shares of The Claremont Company at a price of \($10\) per share, or an aggregate of \($22\) million. Immediately prior to the...
-
Kelly Griffin, an audit manager, had begun preliminary analytical procedures of selected statistics related to the Majestic Hotel. Her objective was to obtain an understanding of the hotel's business...
-
Product A is made from two components, B and C . It takes one B and three Cs to make a single product A . Component B is made from two part Ds . Component C is made from one part D and 2 part Cs ....
-
A firm relies on R&D to maintain profitability. The firm needs to determine the maximum amount to invest today (or invest in phase I) for its three-phased project. Phase I: Research. (Invest R{)...
-
What is the maximum amount of the refundable Child Tax Credit available for each qualifying child age 6 or older in 2023? A) $0. B) $1,600. C) $2,000. D) $3,600.
-
A researcher claims that the percentage of adults in Egypt who own a surround sound speaker is not 40%. Describe type I and type II errors for a hypothesis test of the indicated claim.
-
As stated by a courier companys dispatch department, the number of delivery errors per million consignments has a standard deviation that is less than 2. Write the claim as a mathematical statement....
-
An automotive manufacturer claims that the standard deviation for the gas mileage of one of the vehicles it manufactures is 5.6 kilometers per liter. Determine whether the claim represents the null...
-
Two forecasts were produced, as shown in Table 9.11. The ME for the second forecast is some seven times larger than the ME for the first forecast. However, the MSE is some 20 times larger. Can you...
-
An internet service provider claims that the mean bandwidth drop time is about 7 minutes. You work for one of the ISPs competitors and want to reject the claim. How would you write the null and...
-
how do you put in favourable and unfavorable in an excel spreadsheet ?
-
The tractor is used to lift the 150-kg load B with the 24-mlong rope, boom, and pulley system. If the tractor travels to the right at a constant speed of 4 m/s, determine the tension in the rope when...
-
Use the LE model to describe the bonding in H2CO and C2H2. Carbon is the central atom in H2CO, and C2H2 exists as HCCH.
-
There are two possible structures of XeF2Cl2, where Xe is the central atom. Draw them, and describe how measurements of dipole moments might be used to distinguish among them.
-
What are the two most abundant elements by mass in the earths crust, oceans, and atmosphere? Does this make sense? Why? What are the four most abundant elements by mass in the human body? Does this...
-
The equilibrium for extraction of acetic acid from water into 3heptanol at \(25^{\circ} \mathrm{C}\) is \(\mathrm{y}=0.828 \mathrm{x}\), where \(\mathrm{y}\) is weight fraction acetic acid in...
-
We are extracting acetic acid from water into 3 -heptanol at \(25^{\circ} \mathrm{C}\) in an extraction column with 30 equilibrium stages. Equilibrium is \(y=0.828\) \(\mathrm{x}\), where...
-
A crossflow extraction system is being used to extract furfural from water into methyl-isobutyl ketone (MIBK) at \(25^{\circ} \mathrm{C}\). The \(9000 \mathrm{~kg} / \mathrm{h}\) of aqueous feed...

Study smarter with the SolutionInn App



