Use the diatomic ideal gas heat capacity e-Bhy Cv = R + Nahv G-e-Bhv 1 -...
Fantastic news! We've Found the answer you've been seeking!
Question:

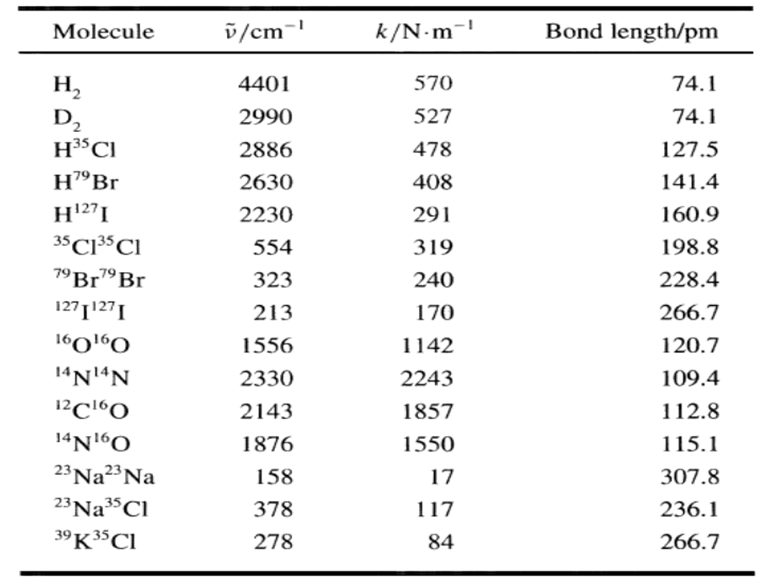
Transcribed Image Text:
Use the diatomic ideal gas heat capacity e-Bhy Cv = R + Nahv G-e-Bhv 1 - e-Bhv Nghv ( a e-Bhy - e-Bhv e-hv/kBT (1 - e-hv/kBT)2 1 hv kBT + and the value of ỹ for HBr given in this table to calculate the equation of the molar heat capacity of HBr in terms of T and R. Cy = II Molecule v/cm-' k/N-m- Bond length/pm 74.1 H, D, 4401 570 2990 527 74.1 H3³CI 2886 478 127.5 H"Br 2630 408 141.4 H27I 2230 291 160.9 35 C13³CI 554 319 198.8 79 Br79 Br 323 240 228.4 1271127I 213 170 266.7 1601ºO 1556 1142 120.7 14N14N 2330 2243 109.4 2143 1857 112.8 14NO 1876 1550 115.1 23 Na23 Na 23 Na3Cl 39K35CI 158 17 307.8 378 117 236.1 278 84 266.7 Use the diatomic ideal gas heat capacity e-Bhy Cv = R + Nahv G-e-Bhv 1 - e-Bhv Nghv ( a e-Bhy - e-Bhv e-hv/kBT (1 - e-hv/kBT)2 1 hv kBT + and the value of ỹ for HBr given in this table to calculate the equation of the molar heat capacity of HBr in terms of T and R. Cy = II Molecule v/cm-' k/N-m- Bond length/pm 74.1 H, D, 4401 570 2990 527 74.1 H3³CI 2886 478 127.5 H"Br 2630 408 141.4 H27I 2230 291 160.9 35 C13³CI 554 319 198.8 79 Br79 Br 323 240 228.4 1271127I 213 170 266.7 1601ºO 1556 1142 120.7 14N14N 2330 2243 109.4 2143 1857 112.8 14NO 1876 1550 115.1 23 Na23 Na 23 Na3Cl 39K35CI 158 17 307.8 378 117 236.1 278 84 266.7
Expert Answer:
Answer rating: 100% (QA)
Answer Solution The wavenumber D for N0 is 1876 cm1 The molar heat capacity Cv is given ... View the full answer

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these accounting questions
-
The molar heat capacity of a diatomic molecule is 29.1 J/K mol. Assuming the atmosphere contains only nitrogen gas and there is no heat loss, calculate the total heat intake (in kilojoules) if the...
-
In Figure where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the cycle with the molecules rotating but not oscillating. What are? (a) p2/p1 (b) p3/p1 and (c) T3/T1 For path 1 ?? 2,...
-
A 1.00-mol sample of a diatomic ideal gas has pressure P and volume V. When the gas is heated, its pressure triples and its volume doubles. This heating process includes two steps, the first at...
-
What is the difference between a Type I error and a Type II error?
-
Based on Figure 1-6 in the text, draw a diagram of functional segments for a manufacturer ofdiversified products. The general characteristics of the firm are as follows: a. The organization produces...
-
Some of the pollutants in the atmosphere are ozone, sulfur dioxide, and sulfur trioxide. Draw Lewis structures for these three molecules. Show all resonance structures.
-
Compute the missing amounts in the following table. (At the end of 2015, retained earnings had a balance of negative $2,086.) Comment on the companys performance over the three-year period after...
-
Diebold, Inc., a public company, is a maker of ATMs, bank security systems, and electronic voting machines. Research how Diebold accounted for liability accruals and capitalized expenses (Division 35...
-
Having each employee record every activity in which they are involved ( as well as the time spent on each activity ) results in a detailed, chronological job analysis collection technique known as...
-
Project 1 is to build a medium-sized garage for a client over two years. The client will pay $10,000 as a deposit immediately, $40,000 the year after and $85,000 in the final year. Your company has a...
-
ion.aspx A Oc. If you deploy appropriate safeguards such as encryption and data masking, data may be stored anywhere in the world. Od. In the internet era, any data may be accessed from anywhere in...
-
Graph f(x) - x, g(x) x +3 and (x) x5. Calculate the derivatives of f, g and h.
-
what ways do contemporary political leaders employ Machiavellian principles to consolidate and maintain power, and what are the ethical ramifications ?
-
Figure Qu. 1 below shows a simple Single Line Diagram (SLD) for a power system where a Utility supplies a 66 kV Customer. - 13.8 kV The 'PG Bus' is a 13.8 kV Power Generation Bus, to which Generators...
-
What is the correct journal entry to prorate the write off of the difference between allocated and actual overhead using the ending balance approach? And What is the correct journal entry to write...
-
Define Least Restrictive Environment What factors should schools consider in placing students in the least restrictive environment?
-
For this problem your code should a. Ask a user to enter in their degree name, e.g. biology or math, input it and store in a variable b. Ask the user the grades they received in their current 4...
-
Nate prepares slides for his microscope. In 1 day he prepared 12 different slides. Which equation best represents y, the total number of slides Nate prepares in x days if he continues at this rate? A...
-
Consider the three formation processes shown for the compound nucleus 20Ne in Figure. Here are some of the masses. What energy must? (a) The alpha particle, (b) The proton, and (c) The -ray photon...
-
The masses and coordinates of three spheres are as follows: 20kg, x = 0.50 m, y = 1.0 m; 40 kg, x = 1.0 m, y = 1.0 m; 60 kg, x = 0m, y = 0.50 m. What is the magnitude of the gravitational force on...
-
In Figure a, a 3.50 g bullet is fired horizontally at two blocks at rest on a frictionless table. The bullet passes through block 1 (mass 1.20 kg) and embeds itself in block 2 (mass 1.80 kg). The...
-
Consider a publicly-held company whose products you are familiar with. Some examples might include: Access the companys public website and search for its most recent annual report. (Note: Some...
-
The 2014 financial statements of LVMH Moet Hennessey-Louis Vuitton S.A. are presented in Appendix C of this book. LVMH is a Paris-based holding company and one of the worlds largest and best-known...
-
San Francisco Sea Salt's controller prepared the following statements of cash flow (in thousands of dollars) for the past two years, the current year (2020), and the upcoming year (2021): After...

Study smarter with the SolutionInn App


